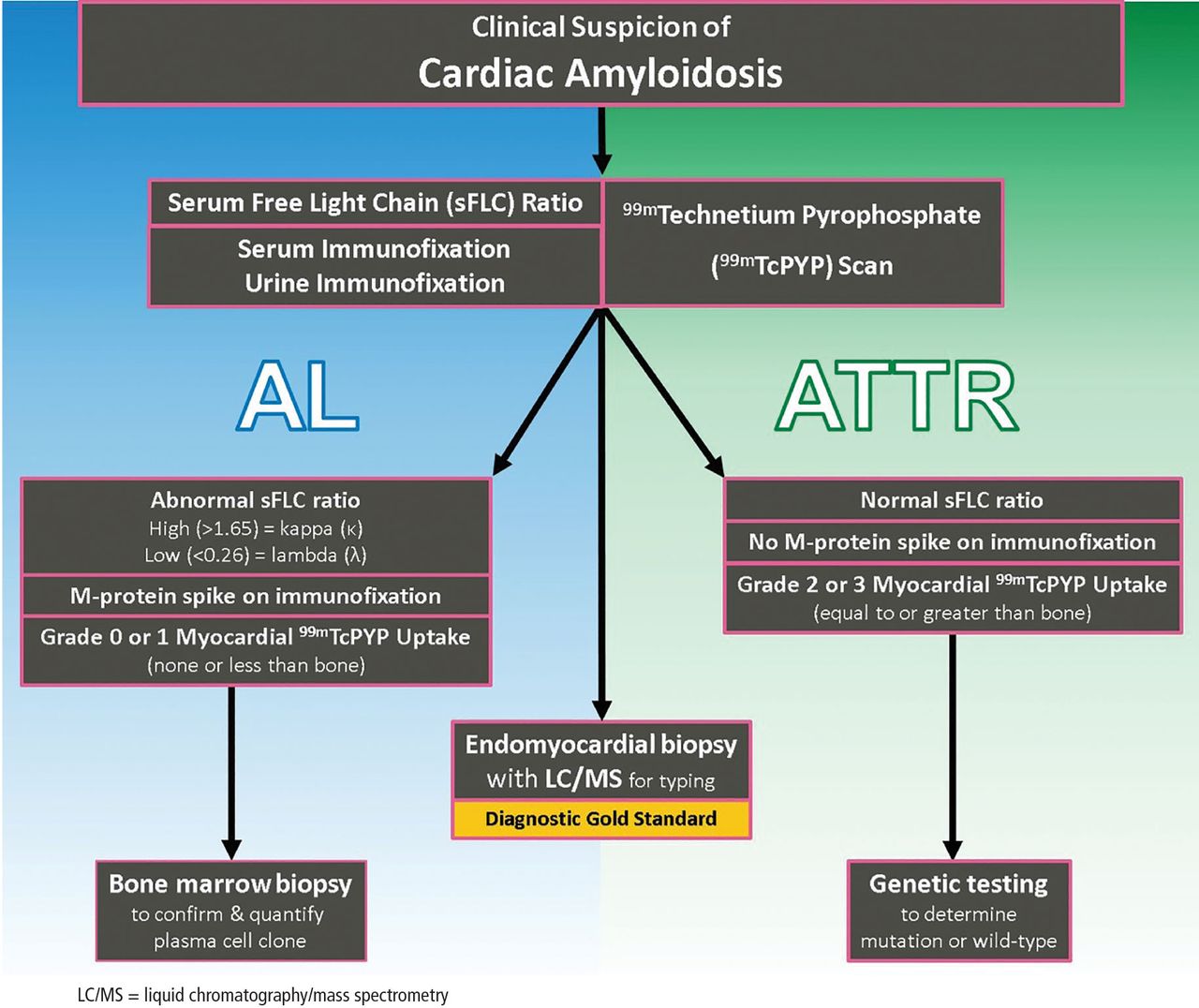
Cardiac Amyloidosis An Update On Diagnosis And Treatment Cleveland Amyloidosis refers to the extracellular deposition of fibrils arising from a variety of serum proteins. these fibrils adopt a beta pleated sheet configuration that leads to characteristic histologic changes [1]. amyloid deposits can occur in a variety of organs, but morbidity and mortality from amyloid deposition most commonly occur when the. J am coll cardiol 2023;jan 23: [epub ahead of print]. the following are key points to remember from a new expert consensus decision pathway document on comprehensive multidisciplinary care for the patient with cardiac amyloidosis: amyloid cardiomyopathy is caused by misfolding of (i) monoclonal immunoglobulin light chain produced in bone marrow.

Pdf Cardiac Amyloidosis An Update On Diagnosis And Treatment As of 2022, tafamidis is the only medication approved by the u.s. food and drug administration (fda) for treatment of attr cardiac amyloidosis. it acts as a ttr stabilizer, slowing the dissociation of ttr and thus fibril formation and cardiac deposition . early diagnosis is crucial because tafamidis delays progression of disease but will not. There are additional cardiac amyloidosis drugs in the pipeline. kittleson said that tafamidis is part of one class of drugs that help stabilize the ttr (transthyretin) protein, preventing it from infiltrating the heart. a second drug in the same class, acoramidis, is currently in phase 3 clinical trials. Until recently, there were zero treatments for nerve or cardiac involved amyloidosis approved by the u.s. food and drug administration (fda). but in 2018, two therapies, inotersen and patisiran, were approved by the fda for hereditary nerve involved attr. and in 2019, tafamidis became the first fda approved drug for hereditary and wild type. Martha grogan, m.d., a cardiologist at mayo clinic who specializes in cardiac amyloidosis, discusses new treatment options.
Treatment Update Medications For Cardiac Amyloidosis Oneamyloidosisvoice Until recently, there were zero treatments for nerve or cardiac involved amyloidosis approved by the u.s. food and drug administration (fda). but in 2018, two therapies, inotersen and patisiran, were approved by the fda for hereditary nerve involved attr. and in 2019, tafamidis became the first fda approved drug for hereditary and wild type. Martha grogan, m.d., a cardiologist at mayo clinic who specializes in cardiac amyloidosis, discusses new treatment options. Cardiac amyloidosis (ca), once thought to be a rare disease, is increasingly recognized due to enhanced clinical awareness and better diagnostic imaging. ca is becoming of heightened interest to the cardiology community given more effective treatment strategies for light chain amyloidosis (al), as well as emerging therapies for transthyretin amyloidosis (attr). A new medication, tafamidis, was approved in 2018 as the first truly disease modifying treatment for attr cardiac amyloidosis, and more medications have been approved for the neuropathy that often results from this disease. “all of these advancements,” vaishnav says, “have led to a lot of excitement in the field.”.

Therapies For Cardiac Light Chain Amyloidosis An Update Cardiac amyloidosis (ca), once thought to be a rare disease, is increasingly recognized due to enhanced clinical awareness and better diagnostic imaging. ca is becoming of heightened interest to the cardiology community given more effective treatment strategies for light chain amyloidosis (al), as well as emerging therapies for transthyretin amyloidosis (attr). A new medication, tafamidis, was approved in 2018 as the first truly disease modifying treatment for attr cardiac amyloidosis, and more medications have been approved for the neuropathy that often results from this disease. “all of these advancements,” vaishnav says, “have led to a lot of excitement in the field.”.
