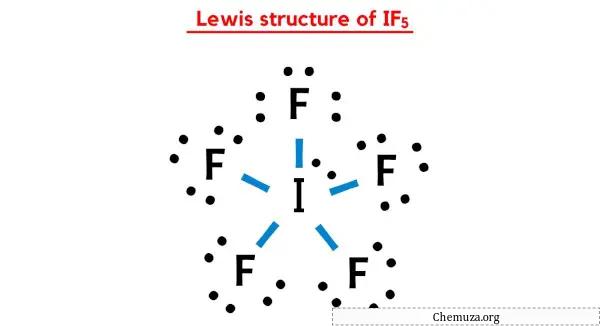
Structure De Lewis If5 En 5 г Tapes Avec Images C La structure if5 lewis a un atome d’iode (i) au centre qui est entouré de cinq atomes de fluor (f). il existe 5 liaisons simples entre l’atome d’iode (i) et chaque atome de fluor (f). il y a 3 paires libres sur tous les atomes de fluor (f) et 1 paire libre sur l’atome d’iode (i). si vous n’avez rien compris de l’image ci dessus. Step 3: connect each atoms by putting an electron pair between them. now in the if5 molecule, you have to put the electron pairs between the iodine atom (i) and fluorine atoms (f). this indicates that the iodine (i) and fluorine (f) are chemically bonded with each other in a if5 molecule. step 4: make the outer atoms stable.
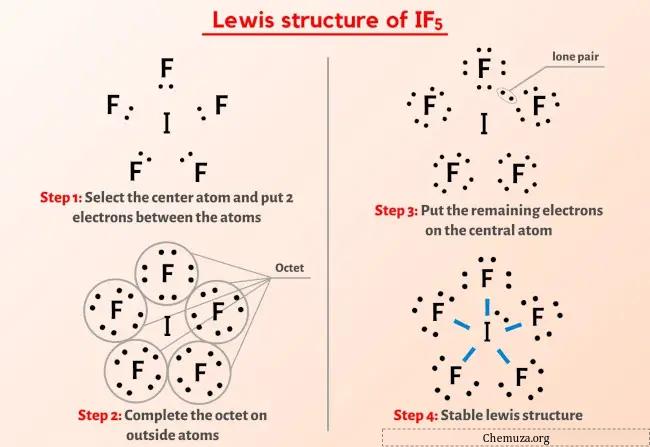
Structure De Lewis If5 En 5 г Tapes Avec Images C Step #1: calculate the total number of valence electrons. here, the given molecule is if5 (iodine pentafluoride). in order to draw the lewis structure of if5, first of all you have to find the total number of valence electrons present in the if5 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). H=6, the molecule will have sp3d2 hybridization. in the case of if5, v = 7 (valence electrons of central atom) m= 5 (5 monovalent atoms of f) since the total charge of if5 is 0, c and a will be zero. hence, h=1 2 [7 5] h=6, indicating that its sp3d2 hybridized. hence, we can easily find the hybridization of if5 using these two methods. Draw an appropriate lewis structure for if5: step 1: identify the central atom: iodine (i) is the central atom in if5 because it's less electronegative than fluorine. step 2: calculate total valence electrons: iodine contributes 7 valence electrons, and each fluorine contributes 7, giving a total of 7 (5 x 7) = 42 valence electrons. step 3. Vous pouvez télécharger le logiciel à l'adresse suivante : acdlabs resources freeware chemsketch.
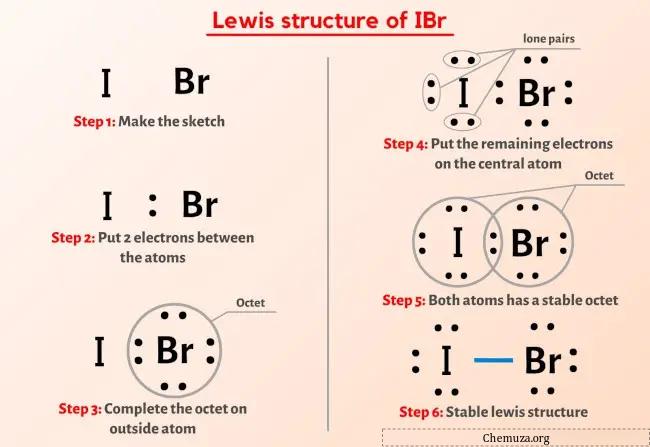
Structure Ibr Lewis En 5 г Tapes Avec Images Chemuza Draw an appropriate lewis structure for if5: step 1: identify the central atom: iodine (i) is the central atom in if5 because it's less electronegative than fluorine. step 2: calculate total valence electrons: iodine contributes 7 valence electrons, and each fluorine contributes 7, giving a total of 7 (5 x 7) = 42 valence electrons. step 3. Vous pouvez télécharger le logiciel à l'adresse suivante : acdlabs resources freeware chemsketch. If5 lewis structure. the information on this page is fact checked. if 5 (iodine pentafluoride) has one iodine atom and five fluorine atoms. in the if 5 lewis structure, there are five single bonds around the iodine atom, with five fluorine atoms attached to it. each fluorine atom has three lone pairs, and the iodine atom has one lone pair. First, we need to draw the lewis structure of if5. in short, these are the steps you need to follow for drawing a lewis structure: 1. write the correct skeletal structure for the molecule. * hydrogen atoms are always terminal (only one bond) * put more electronegative elements in terminal positions. 2. sum the valence electrons from all the atoms.
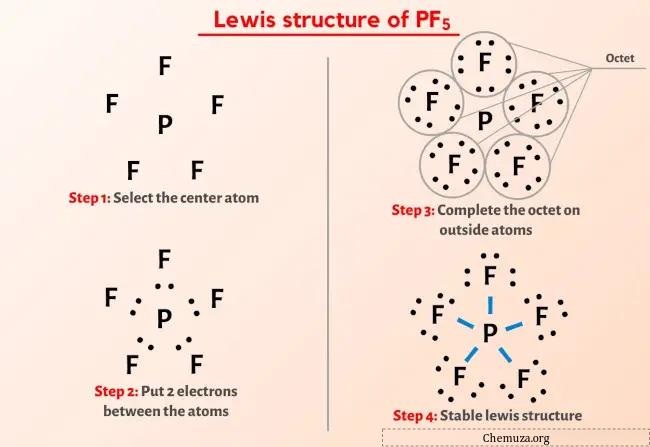
Structure De Lewis Pf5 En 5 г Tapes Avec Images Chemuza If5 lewis structure. the information on this page is fact checked. if 5 (iodine pentafluoride) has one iodine atom and five fluorine atoms. in the if 5 lewis structure, there are five single bonds around the iodine atom, with five fluorine atoms attached to it. each fluorine atom has three lone pairs, and the iodine atom has one lone pair. First, we need to draw the lewis structure of if5. in short, these are the steps you need to follow for drawing a lewis structure: 1. write the correct skeletal structure for the molecule. * hydrogen atoms are always terminal (only one bond) * put more electronegative elements in terminal positions. 2. sum the valence electrons from all the atoms.
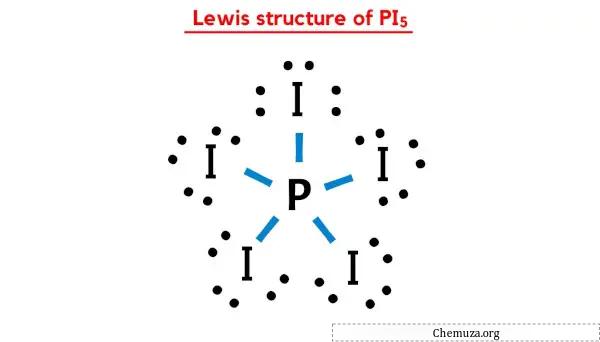
Structure Pi5 Lewis En 5 г Tapes Avec Images Chemuza
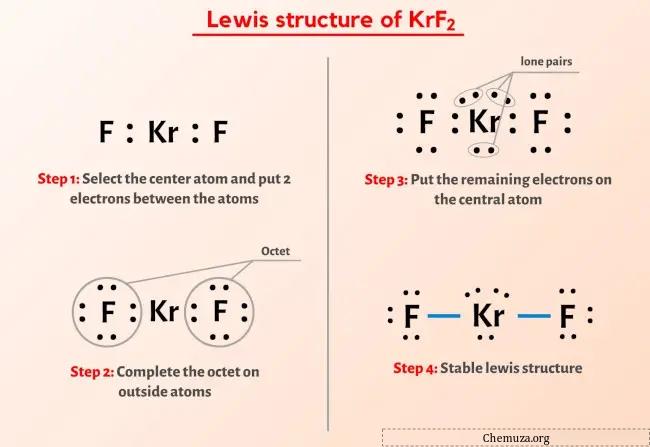
Structure De Lewis Krf2 En 5 г Tapes Avec Images Chemuz
