Solved Draw The Lewis Structure Of Methane Ch4 Which Vrogue Co A step by step explanation of how to draw the ch4 lewis dot structure (methane).for the ch4 structure use the periodic table to find the total number of vale. Craig beals shows how to draw the lewis structure for methane.this is a clip from the complete video: covalent bonding 2.1 drawing lewis structures.
Solved Draw The Lewis Structure Of Methane Ch4 Which Vrogue Co A video explanation of how to draw the lewis dot structure for methane, along with information about the compound including formal charges, polarity, hybrid. Science. chemistry. chemistry questions and answers. draw and reference the lewis structure for methane, ch4, then answer the questions. 1. how many total valence electrons are counted in the lewis structure for methane? [ select) 2. how many bonding electron pairs are counted?. Drawing the lewis structure for ch 4. for ch 4 you have a total of 8 total valence electrons. drawing the lewis structure for ch 4 (named methane) requires only single bonds. it's one of the easier lewis structures to draw. remember that hydrogen atoms always go on the outside of a lewis structure and that they only need two valence electrons. Chemistry questions and answers. draw the lewis structure for methane (ch4). how many σ bonds and pi bonds are there in the entire molecule? four σ bonds and zero pi bonds four σ bonds and one pi bond four σ bonds and two pi bonds three σ bonds and one pi bond two σ bonds and two pi bonds one σ bond and three pi bonds one σ bond and two.

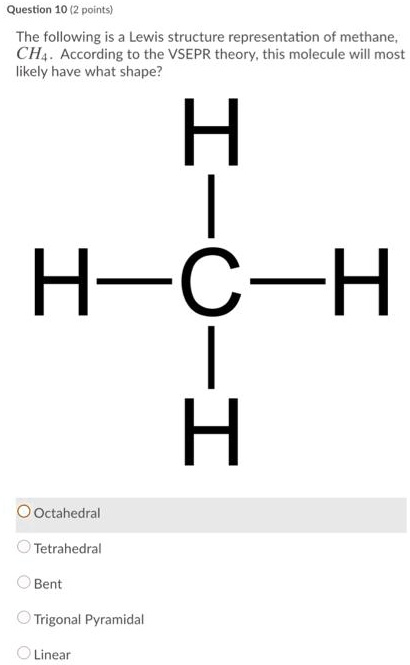
Solved Draw The Lewis Structure For Methane Ch4 And E Vrogueо Drawing the lewis structure for ch 4. for ch 4 you have a total of 8 total valence electrons. drawing the lewis structure for ch 4 (named methane) requires only single bonds. it's one of the easier lewis structures to draw. remember that hydrogen atoms always go on the outside of a lewis structure and that they only need two valence electrons. Chemistry questions and answers. draw the lewis structure for methane (ch4). how many σ bonds and pi bonds are there in the entire molecule? four σ bonds and zero pi bonds four σ bonds and one pi bond four σ bonds and two pi bonds three σ bonds and one pi bond two σ bonds and two pi bonds one σ bond and three pi bonds one σ bond and two. Step #1: calculate the total number of valence electrons. here, the given molecule is ch4 (methane). in order to draw the lewis structure of ch4, first of all you have to find the total number of valence electrons present in the ch4 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). The lewis structure of methane (ch3) is as follows: {{{ h | h c h | h {{{ methane's molecular structure is tetrahedral. for methane, the abe formula would be ax₄, indicating that four atoms (hydrogens) are linked to the central carbon atom. abe stands for the central atom, b for the surrounding atoms, and e for the number of lone pairs.

Solved Draw The Lewis Structure For Methane Ch4 And E Vrogueо Step #1: calculate the total number of valence electrons. here, the given molecule is ch4 (methane). in order to draw the lewis structure of ch4, first of all you have to find the total number of valence electrons present in the ch4 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). The lewis structure of methane (ch3) is as follows: {{{ h | h c h | h {{{ methane's molecular structure is tetrahedral. for methane, the abe formula would be ax₄, indicating that four atoms (hydrogens) are linked to the central carbon atom. abe stands for the central atom, b for the surrounding atoms, and e for the number of lone pairs.
