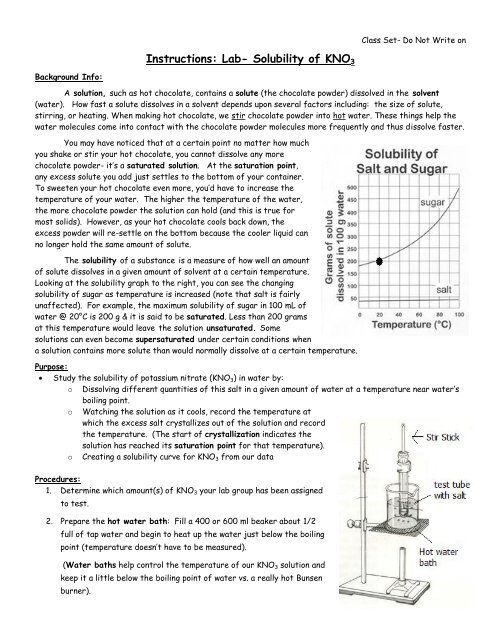
Instructions Lab Solubility Of Kno3 The reaction to be studied is the solubility of kno3(s). kno3(s) ↔ k (aq) no3 (aq) let s dissolve s s. k = ksp = [k ] [no3 ] = s2. the source of your data is the solubility graph below. (you will need to convert the solubility units given in the graph to molarity for the calculation of the equilibrium constant. 79, 77, 80. 81, 80, 75, 79. purpose: find crystallization temperatures for 7 concentrations of kno3 and make a solubility graph materials: kno3, test tube, stir rod, weigh boats, hot plates, thermometer, 10 ml graduated cylinder. procedure: in two test tubes, put exactly 5 ml water in each put assigned amount kno3 in each heat in hot water.
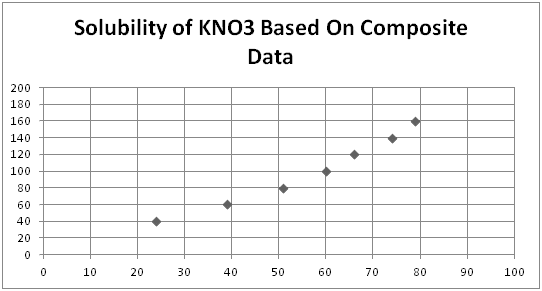
Solubility Of Kno3 Lab Table Graph Schoolworkhelper Namics of potassium nitrate dissolving in water version v121113objectivethe Δg, Δh and Δs of the potassium nitrate (kno3) dissolving reaction will be deter. ri. g the equilibrium constant (ksp) at different temperatures. background1. solubility product constant (. in a saturated potassium nitrate (kno3) solution in water (h2o), a dynamic. Instructions: lab solubility of kno 3 background info: a solution, such as hot chocolate, contains a solute (the chocolate powder) dissolved in the solvent (water). how fast a solute dissolves in a solvent depends upon several factors including: the size of solute, stirring, or heating. when making hot chocolate, we stir chocolate powder into. Then plug in y, the solubility: a) (35 g)*2 100 ml = 87 g 100 ml when y = 87, the temperature, x ≈ 60°c b) (20 g)*2 100 ml = 50 g 100 ml when y = 50, the temperature, x ≈ 42°c 5. a) if the solubility is above the curve on the graph, the solution is supersaturated. b) if the solubility is on the curve on the graph, the solution is saturated. You will determine three thermodynamic values associated with the solubility of potassium nitrate. the reaction we will be studying is the dissolution of potassium nitrate in water. kno3(s) ↽−−⇀ k (aq) no−3 (aq) (11.1) (11.1) kno 3 (s) ↽ − − ⇀ k (a q) no 3 − (a q) potassium nitrate is a highly soluble compound (large.
Chemistry Lab Lab 12 Solubility Of Kno3 Then plug in y, the solubility: a) (35 g)*2 100 ml = 87 g 100 ml when y = 87, the temperature, x ≈ 60°c b) (20 g)*2 100 ml = 50 g 100 ml when y = 50, the temperature, x ≈ 42°c 5. a) if the solubility is above the curve on the graph, the solution is supersaturated. b) if the solubility is on the curve on the graph, the solution is saturated. You will determine three thermodynamic values associated with the solubility of potassium nitrate. the reaction we will be studying is the dissolution of potassium nitrate in water. kno3(s) ↽−−⇀ k (aq) no−3 (aq) (11.1) (11.1) kno 3 (s) ↽ − − ⇀ k (a q) no 3 − (a q) potassium nitrate is a highly soluble compound (large. 1 answer. the only way to determine the solubility of potassium nitrate is to do an experiment or interpolate from a solubility graph. solubility is the mass of a solute that can be dissolved in a given mass of solvent. solubility varies according to temperature for the same solute and solvent. the solubility graph for potassium nitrate in. In each measurement you mix known masses of the salt and the water. problem 1: figure 2.2.2 2.2. 2 (a) is reading the temperature of a saturated solution, the problem is we do not know the concentration of the salt that dissolved. that is, the total salt added is the mass of the salt dissolved and the mass of the precipitate.
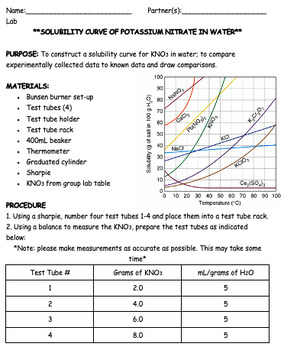
Solubility Curves Of Kno3 Lab By Science And Sass In Miss G S Class 1 answer. the only way to determine the solubility of potassium nitrate is to do an experiment or interpolate from a solubility graph. solubility is the mass of a solute that can be dissolved in a given mass of solvent. solubility varies according to temperature for the same solute and solvent. the solubility graph for potassium nitrate in. In each measurement you mix known masses of the salt and the water. problem 1: figure 2.2.2 2.2. 2 (a) is reading the temperature of a saturated solution, the problem is we do not know the concentration of the salt that dissolved. that is, the total salt added is the mass of the salt dissolved and the mass of the precipitate.
