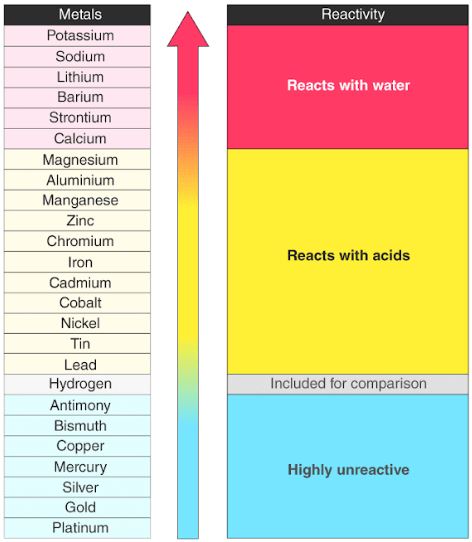
Reactivity Series Reactivity Of Metals Chart Features Uses The data provided by the reactivity series can be used to predict whether a metal can displace another in a single displacement reaction. it can also be used to obtain information on the reactivity of metals towards water and acids. 3,093. a chart of the reactivity series of common metals is provided below. metals tend to readily lose electrons. Learn how to use the activity series of metals or reactivity series to predict chemical reactions. see a chart of metals from most to least reactive and examples of how to apply the series.

Csec Chemistry Reactivity Of Metals The activity series is a list of elements in decreasing order of their reactivity. since metals replace other metals, while nonmetals replace other nonmetals, they each have a separate activity series. the table 7.11.1 below is an activity series of most common metals, and the table 7.11.2 is an activity series of the halogens. Learn what the reactivity series of metals is, how it is arranged, and why it is important. see examples of reactions of metals with water, acids, and salts based on the reactivity series. Reactivity series. in chemistry, a reactivity series (or reactivity series of elements) is an empirical, calculated, and structurally analytical progression [1] of a series of metals, arranged by their "reactivity" from highest to lowest. [2][3][4] it is used to summarize information about the reactions of metals with acids and water, single. P3: activity series of metals. the reactivity series is a series of metals, in order of reactivity from highest to lowest. it is used to determine the products of single displacement reactions, whereby metal a will replace another metal b in a solution if a is higher in the series. activity series of some of the more common metals, listed in.

The Reactivity Series Revision Notes In Gcse Chemistry Reactivity series. in chemistry, a reactivity series (or reactivity series of elements) is an empirical, calculated, and structurally analytical progression [1] of a series of metals, arranged by their "reactivity" from highest to lowest. [2][3][4] it is used to summarize information about the reactions of metals with acids and water, single. P3: activity series of metals. the reactivity series is a series of metals, in order of reactivity from highest to lowest. it is used to determine the products of single displacement reactions, whereby metal a will replace another metal b in a solution if a is higher in the series. activity series of some of the more common metals, listed in. The activity series is a chart of metals listed in order of declining relative reactivity. the top metals are more reactive than the metals on the bottom. for example, both magnesium and zinc can react with hydrogen ions to displace h 2 from a solution by the reactions: mg (s) 2 h (aq) → h 2 (g) mg 2 (aq) zn (s) 2 h (aq) → h 2 (g. The reactivity series is a key concept in chemistry that explains elemental behavior and its role in chemical processes. it is a useful instrument in the area of chemical research, whether it’s anticipating reaction outcomes, understanding metal extraction methods, or implementing corrosion prevention measures.
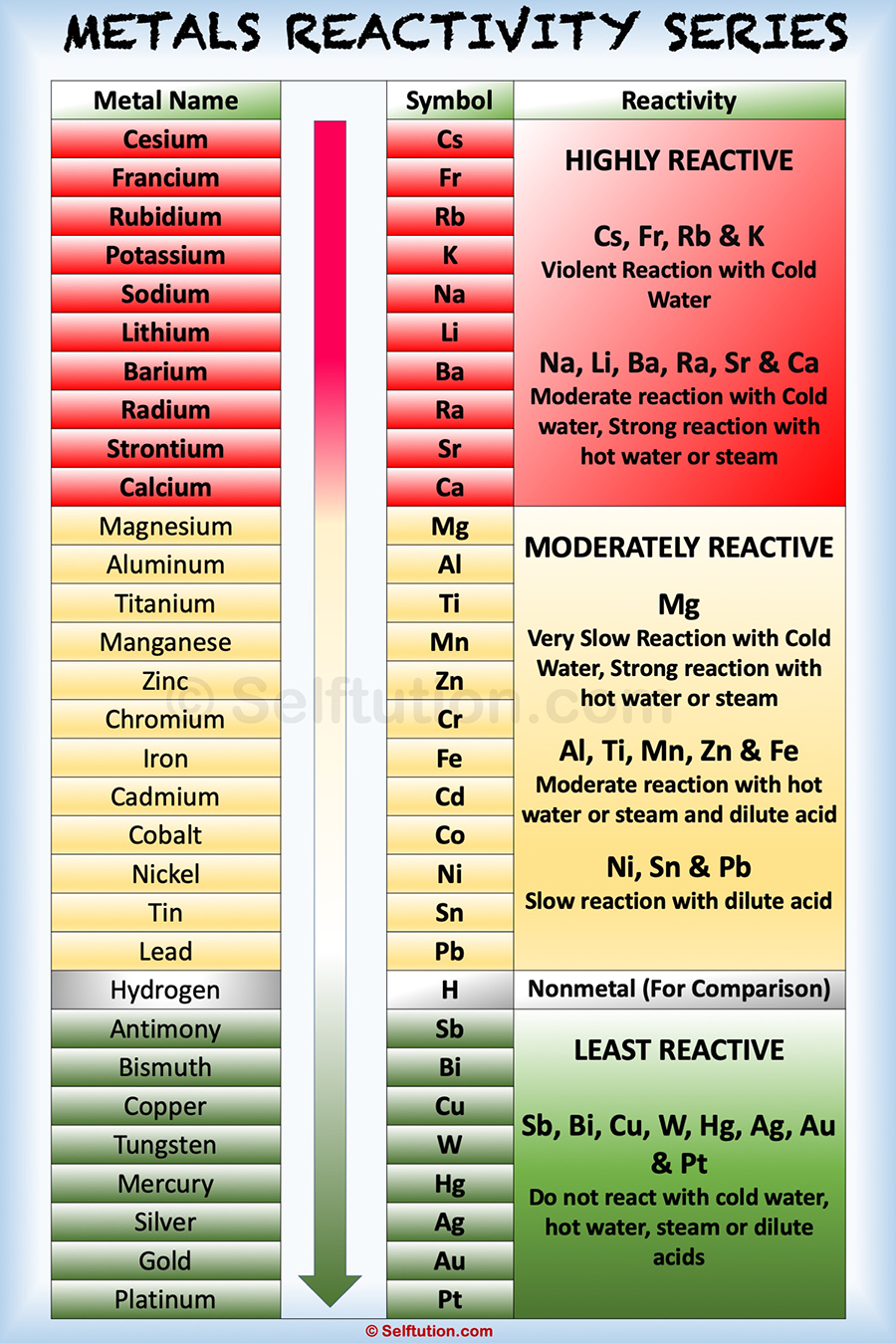
Reactivity Series Of Metals And Nonmetals в Selftution The activity series is a chart of metals listed in order of declining relative reactivity. the top metals are more reactive than the metals on the bottom. for example, both magnesium and zinc can react with hydrogen ions to displace h 2 from a solution by the reactions: mg (s) 2 h (aq) → h 2 (g) mg 2 (aq) zn (s) 2 h (aq) → h 2 (g. The reactivity series is a key concept in chemistry that explains elemental behavior and its role in chemical processes. it is a useful instrument in the area of chemical research, whether it’s anticipating reaction outcomes, understanding metal extraction methods, or implementing corrosion prevention measures.
