
Oxidation And Reduction Of Alkenes And Alkynes Mcat And Organic Alkenes undergo a number of reactions in which the c=c double bond is oxidized. for organic compounds, a conventional way to tell whether the oxidation or reduction occur is to check the number of c–o bonds or the c–h bonds. an oxidation reaction increase the number of c–o bonds or decrease the number of c–h bonds. 10.7 oxidation reactions of alkenes. alkenes undergo a number of reactions in which the c=c double bond is oxidized. for organic compounds, a conventional way to tell whether oxidation or reduction occurs is to check the number of c–o bonds or c–h bonds. an oxidation reaction increases the number of c–o bonds or decreases the number of c.
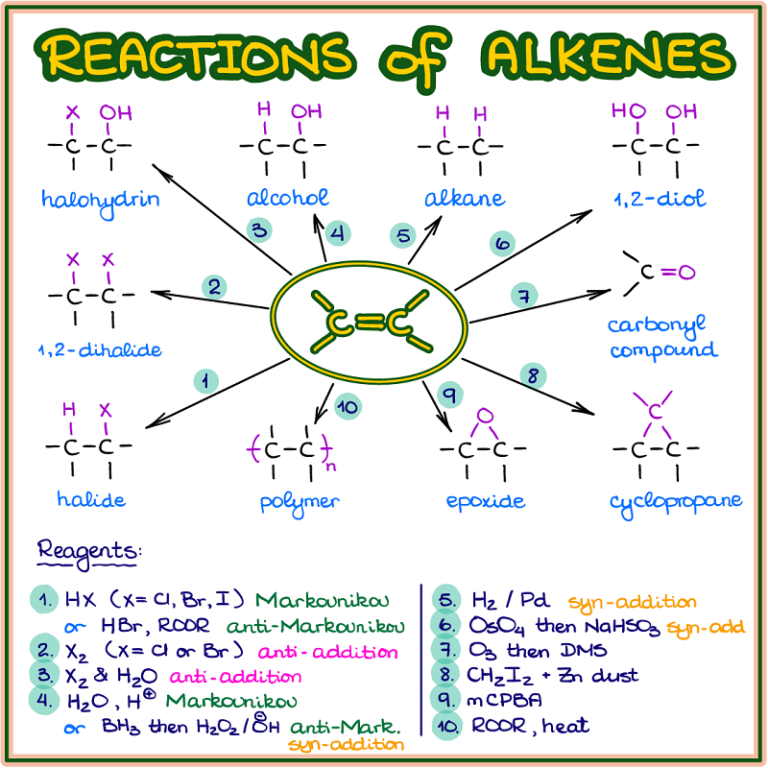
Reactions Of Alkenes вђ Organic Chemistry Tutor Dihydroxylation of alkenes. alkenes are oxidized to cis 1,2 diols by osmium tetroxide (oso4). the stereospecificity is due to the formation of a cyclic osmate ester intermediate. osmium tetroxide can be used directly, but it is normally used in catalytic amounts, and is regenerated by n methylmorpholine n oxide. 8.7 • oxidation of alkenes: epoxidation and hydroxylation like the word reduction used in the previous section for the addition of hydrogen to a double bond, the word oxidation has a slightly different meaning in organic chemistry than what you might have previously learned. This page titled 11.7: oxidation reactions is shared under a cc by nc sa 4.0 license and was authored, remixed, and or curated by john d. roberts and marjorie c. caserio. most alkenes react readily with ozone, even at low temperatures, to yield cyclic peroxidic derivatives known as ozonides. ozonization of alkenes has been studied extensively. Epoxidation of alkenes. alkenes can be oxidized to epoxides using a ‘ peroxyacid ‘ such as m chloroperoxybenzoic acid (mcpba). notice the presence of a third oxygen in the peroxyacid functional group. the mechanism was covered earlier in section 10.7. the π electrons in the alkene double bond attacking the ‘outer’ oxygen of the.

Alkenes Oxidation And Reductions Reactions Youtube This page titled 11.7: oxidation reactions is shared under a cc by nc sa 4.0 license and was authored, remixed, and or curated by john d. roberts and marjorie c. caserio. most alkenes react readily with ozone, even at low temperatures, to yield cyclic peroxidic derivatives known as ozonides. ozonization of alkenes has been studied extensively. Epoxidation of alkenes. alkenes can be oxidized to epoxides using a ‘ peroxyacid ‘ such as m chloroperoxybenzoic acid (mcpba). notice the presence of a third oxygen in the peroxyacid functional group. the mechanism was covered earlier in section 10.7. the π electrons in the alkene double bond attacking the ‘outer’ oxygen of the. Alkenes are much more reactive toward catalytic hydrogenation than most other unsaturated functional groups, and the reaction is therefore quite selective. other functional groups, such as aldehydes, ketones, esters, and nitriles, often survive alkene hydrogenation conditions unchanged, although reaction with these groups does occur under more. Leah4sci redox presents: oxidation, oxidative cleavage, and reduction reactions for alkenes and alkynes.need help with orgo? download my free guide '10 s.
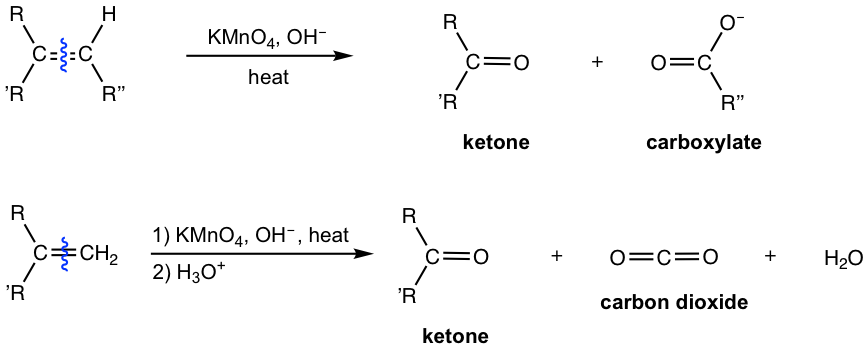
10 7 Oxidation Reactions Of Alkenes вђ Organic Chemistry Alkenes are much more reactive toward catalytic hydrogenation than most other unsaturated functional groups, and the reaction is therefore quite selective. other functional groups, such as aldehydes, ketones, esters, and nitriles, often survive alkene hydrogenation conditions unchanged, although reaction with these groups does occur under more. Leah4sci redox presents: oxidation, oxidative cleavage, and reduction reactions for alkenes and alkynes.need help with orgo? download my free guide '10 s.
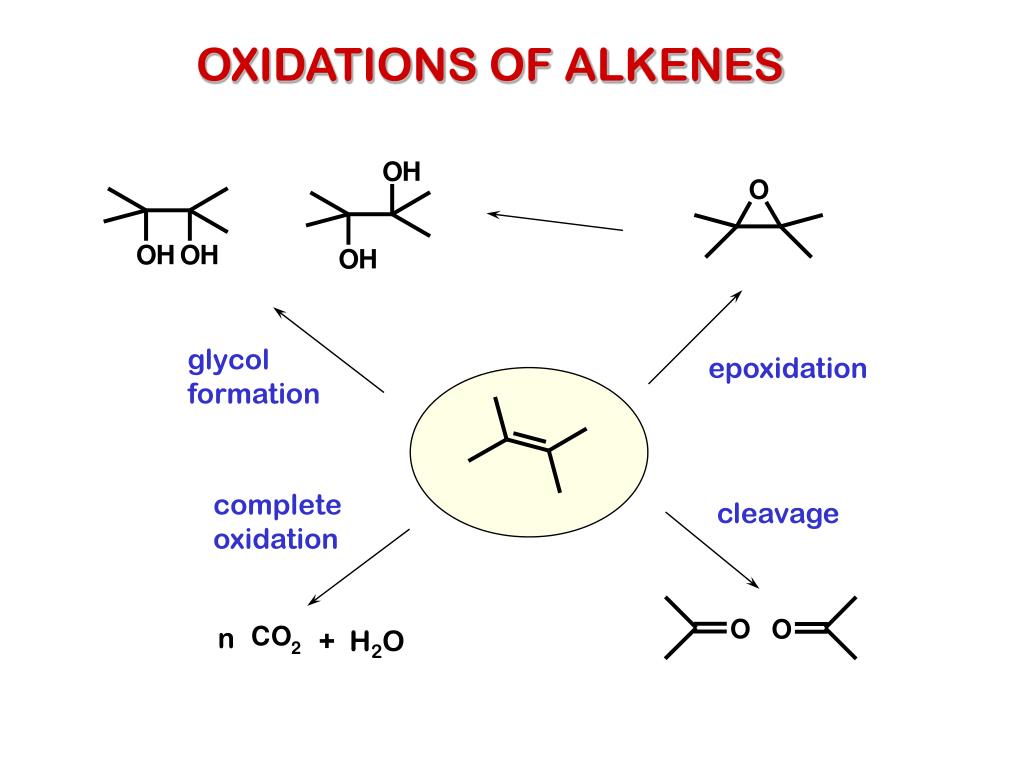
Ppt Oxidation And Reduction Of Alkenes Powerpoint Presentation Free
