
Na2co3 Ch3cooh Ch3coona Nahco3 Chemical Reaction And Equation вђ Otosection Step 4: substitute coefficients and verify result. count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges ions) are balanced. na2co3 2 ch3cooh = 2 ch3coona co2 h2o. reactants. Step 4: substitute coefficients and verify result. count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges ions) are balanced. 3 ch3cooh 2 nahco3 = 2 ch3coona 4 h2o 4 co. reactants.

Na2co3 Co2 H2o Nahco3 Chemical Reaction And Equation There are three main steps for writing the net ionic equation for na2co3 ch3cooh = nach3coo co2 h2o (sodium carbonate acetic acid). first, we balance. Instructions on balancing chemical equations: enter an equation of a chemical reaction and click 'balance'. the answer will appear below; always use the upper case for the first character in the element name and the lower case for the second character. examples: fe, au, co, br, c, o, n, f. compare: co cobalt and co carbon monoxide. Solved and balanced chemical equation nahco3 ch3cooh → h2o co2 ch3coona with completed products. application for completing products and balancing equations. Half of this is a double displacement reaction. the reaction takes place in two steps. step 1 ch₃cooh(aq) nahco₃(s) → ch₃coona h₂co₃(aq) this is a double displacement reaction because the na trades partners with the h of ch₃cooh. step 2 the carbonic acid formed in step 1 decomposes into co₂ and water. h₂co₃(aq) → co₂(g) h₂o(l) this is a decomposition reaction.

How To Write The Net Ionic Equation For Na2co3 Ch3cooh Nach3coo Solved and balanced chemical equation nahco3 ch3cooh → h2o co2 ch3coona with completed products. application for completing products and balancing equations. Half of this is a double displacement reaction. the reaction takes place in two steps. step 1 ch₃cooh(aq) nahco₃(s) → ch₃coona h₂co₃(aq) this is a double displacement reaction because the na trades partners with the h of ch₃cooh. step 2 the carbonic acid formed in step 1 decomposes into co₂ and water. h₂co₃(aq) → co₂(g) h₂o(l) this is a decomposition reaction. Step 4: substitute coefficients and verify result. count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges ions) are balanced. 8 naco3 9 ch3cooh = 8 ch3coona 10 co2 6 h2o. reactants. Instructions on balancing chemical equations: enter an equation of a chemical reaction and click 'balance'. the answer will appear below; always use the upper case for the first character in the element name and the lower case for the second character. examples: fe, au, co, br, c, o, n, f. compare: co cobalt and co carbon monoxide.
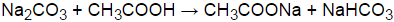
Na2co3 Ch3cooh Ch3coona Nahco3 р рµр рєс рёсџ рір р рёрјрѕрґрµр сѓс рірёсџ р Step 4: substitute coefficients and verify result. count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges ions) are balanced. 8 naco3 9 ch3cooh = 8 ch3coona 10 co2 6 h2o. reactants. Instructions on balancing chemical equations: enter an equation of a chemical reaction and click 'balance'. the answer will appear below; always use the upper case for the first character in the element name and the lower case for the second character. examples: fe, au, co, br, c, o, n, f. compare: co cobalt and co carbon monoxide.
