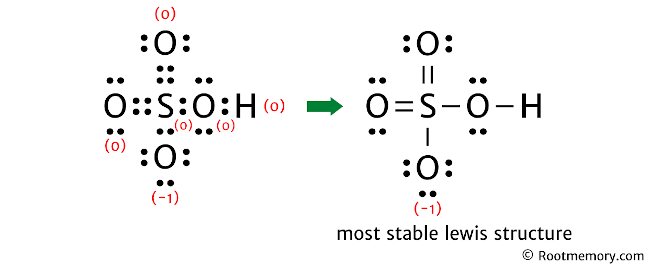
Lewis Structure Of Hso4 Root Memory The lewis structure of hso4– contains two double bonds and three single bonds, with sulfur in the center, and hydrogen and four oxygens on either side. the top oxygen atom, left oxygen atom, and right oxygen atom have two lone pairs. the bottom oxygen atom has three lone pairs, and sulfur atom and hydrogen atom do not have any lone pair. The information on this page is fact checked. lewis structure of h 2 so 4 | image: root memory. the lewis structure of h2so4 contains two double bonds and four single bonds, with sulfur in the center, and two hydrogens and four oxygens on either side. there are two lone pairs on each oxygen atom, and sulfur atom and hydrogen atom do not have.
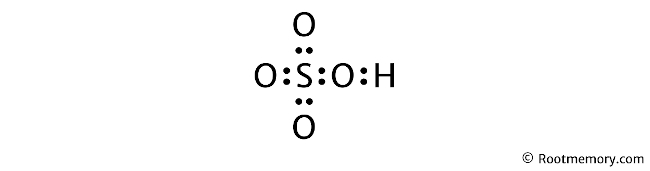
Lewis Structure Of Hso4 Root Memory A step by step explanation of how to draw the hso4 lewis dot structure (bisulfate ion or hydrogen sulfate ion).when we have an h (or h2) in front of a polya. Step #1: calculate the total number of valence electrons. here, the given molecule is hso4 ion (hydrogen sulfate ion). in order to draw the lewis structure of hso4 , first of all you have to find the total number of valence electrons present in the hso4 ion. (valence electrons are the number of electrons present in the outermost shell of an. 🚀to book a personalized 1 on 1 tutoring session:👉janine the tutor janinethetutor 🚀more proven oneclass services you might be interested in:👉one. Your instructions for only $21 task. learn more. sulfuric acid, with the chemical formula h2so4, consists of two hydrogen atoms (h), one sulfur atom (s), and four oxygen atoms (o). the lewis structure of h2so4 allows us to visualize the arrangement of atoms, the bonding patterns, and the distribution of valence electrons within the molecule.
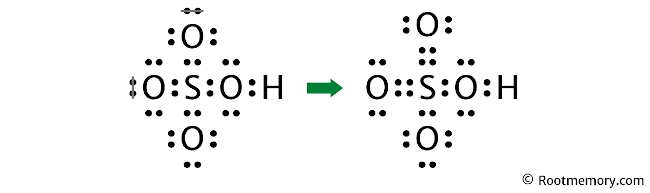
Lewis Structure Of Hso4 Root Memory 🚀to book a personalized 1 on 1 tutoring session:👉janine the tutor janinethetutor 🚀more proven oneclass services you might be interested in:👉one. Your instructions for only $21 task. learn more. sulfuric acid, with the chemical formula h2so4, consists of two hydrogen atoms (h), one sulfur atom (s), and four oxygen atoms (o). the lewis structure of h2so4 allows us to visualize the arrangement of atoms, the bonding patterns, and the distribution of valence electrons within the molecule. Draw the lewis structure for h2so4, hso4–, and so42–. for each species, determine the maximum number of equivalent resonance structures. sulfur is the central atom in all three species. on the other hand, if the molecule contains hydrogen atoms they are attached to the oxygen atoms. only include the best structures, e.g. a structure with bad formal charges should not be included. the. The lewis electron structure for the nh 4 ion is as follows: the nitrogen atom shares four bonding pairs of electrons, and a neutral nitrogen atom has five valence electrons. using equation 8.5.1, the formal charge on the nitrogen atom is therefore. formal charge(n) = 5 − (0 8 2) = 0.
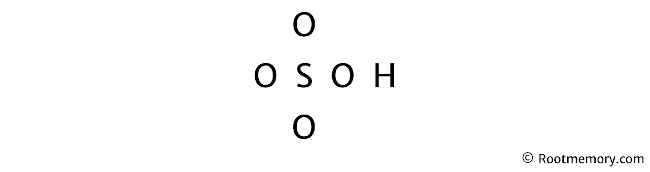
Lewis Structure Of Hso4 Root Memory Draw the lewis structure for h2so4, hso4–, and so42–. for each species, determine the maximum number of equivalent resonance structures. sulfur is the central atom in all three species. on the other hand, if the molecule contains hydrogen atoms they are attached to the oxygen atoms. only include the best structures, e.g. a structure with bad formal charges should not be included. the. The lewis electron structure for the nh 4 ion is as follows: the nitrogen atom shares four bonding pairs of electrons, and a neutral nitrogen atom has five valence electrons. using equation 8.5.1, the formal charge on the nitrogen atom is therefore. formal charge(n) = 5 − (0 8 2) = 0.

Hso4 Lewis Structure How To Draw The Lewis Structure For вђ Otosection
