
Hbr Lewis Structure A step by step explanation of how to draw the hbr lewis dot structure (hydrogen bromide).for the hbr structure use the periodic table to find the total numbe. Step 1. to find out the lewis structure of any given molecule, the first step is to find out the total valence electron number. electron loss signifies the increase of positive charge hence we use the sign ‘ ’. the gain of electrons increases the number of negatively charged electrons therefore we use the ‘ ’ sign.
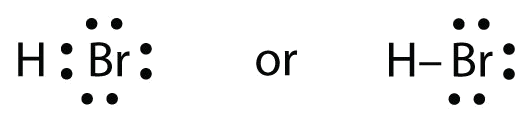
Kbr Lewis Structure The hbr lewis structure represents the arrangement of atoms and bonding electrons in a molecule of hydrogen bromide (hbr). it showcases the connectivity between hydrogen (h) and bromine (br) atoms, giving us a visual representation of their covalent bond. atomic information: hydrogen (h): 1 proton, 1 electron bromine (br): 35 protons, 35 electrons drawing the. Drawing the lewis structure for hbr. hbr is very similar to hf and hcl. hydrogen has 1 valence electron and br (in group 7 with f and cl) has 7 valence electrons. with the lewis structure for hbr remember that hydrogen only needs 2 valence electrons to have a full outer shell. be sure that you don't use more than the 8 valence electrons available. Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Lewis structure finder. added jun 9, 2014 by webtester in chemistry. this widget gets the lewis structure of chemical compounds. send feedback | visit wolfram|alpha. and follow the easy directions provided by blogger. on the next page click the "add" button. you will then see the widget on your igoogle account.
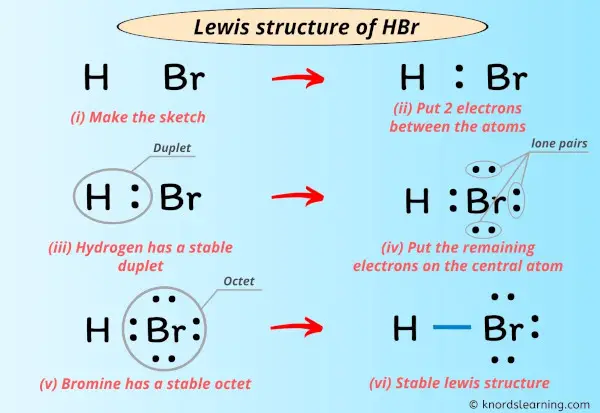
Lewis Structure Of Hbr With 6 Simple Steps To Draw Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Lewis structure finder. added jun 9, 2014 by webtester in chemistry. this widget gets the lewis structure of chemical compounds. send feedback | visit wolfram|alpha. and follow the easy directions provided by blogger. on the next page click the "add" button. you will then see the widget on your igoogle account. In the above structure, you can see that the central atom (bromine) forms an octet. and the outside atom (hydrogen) also forms a duet. hence, the octet rule and duet rule are satisfied. therefore, this structure is the stable lewis structure of hbr. next: n 2 h 4 lewis structure your feedback matters. visit our contact page. What is the lewis dot structure for hydrogen bromide (hbr)? the total number of electrons would 8. there is a single bond connecting hydrogen and bromine. since hydrogen is satisfied by only two electrons, the rest of the lone pairs will end up on bromine. what is the lewis dot structure of carbon tetrachloride (ccl 4)?.
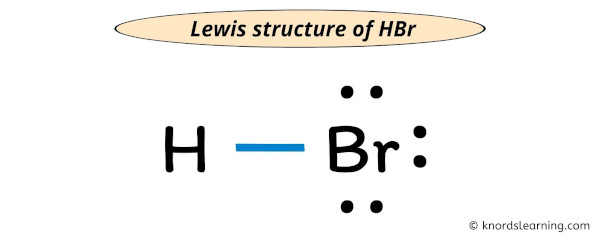
Lewis Structure Of Hbr With 6 Simple Steps To Draw In the above structure, you can see that the central atom (bromine) forms an octet. and the outside atom (hydrogen) also forms a duet. hence, the octet rule and duet rule are satisfied. therefore, this structure is the stable lewis structure of hbr. next: n 2 h 4 lewis structure your feedback matters. visit our contact page. What is the lewis dot structure for hydrogen bromide (hbr)? the total number of electrons would 8. there is a single bond connecting hydrogen and bromine. since hydrogen is satisfied by only two electrons, the rest of the lone pairs will end up on bromine. what is the lewis dot structure of carbon tetrachloride (ccl 4)?.
