
How To Find The Oxidation Number For P In The Po2 3 Ion To find the correct oxidation state of p in po2 3 (the hypophosphite ion), and each element in the ion, we use a few rules and some simple math.first, since. Enter the formula of a chemical compound to find the oxidation number of each element. a net ionic charge can be specified at the end of the compound between { and }. for example: zncl4 {2 } or nh2nh3 { }. enter just an element symbol to show the common and uncommon oxidation states of the element. use uppercase for the first character in the.

How To Find The Oxidation Number For P In P2o3 Youtube To find the correct oxidation state of p in po3 3 (the phosphite ion), and each element in the ion, we use a few rules and some simple math.first, since the. Be on the lookout for any exceptional cases for o, h, etc. in na 2 so 4, we know, based on our set of rules, that the na ion has a charge (and thus oxidation number) of 1 and that the oxygen atoms have oxidation numbers of 2. 3. multiply the number of each atom by its oxidation number. To calculate oxidation numbers of elements in the chemical compound, enter it's formula and click 'calculate' (for example: ca2 , hf2^ , fe4 [fe (cn)6]3, nh4no3, so42 , ch3cooh, cuso4*5h2o). the oxidation state of an atom is the charge of this atom after ionic approximation of its heteronuclear bonds. the oxidation number is synonymous with the. The sum of the oxidation numbers in a polyatomic ion is equal to the charge on that ion. again, work backwards to determine the oxidation number of any non oxygen or non hydrogen atom. to determine the oxidation number of cr in cr 2 o 7 2 : oxygen will be 2 (rule 4), for a total of: 2 × 7 = 14.

Oxidation Number State Definition Rules How To Find And Examples To calculate oxidation numbers of elements in the chemical compound, enter it's formula and click 'calculate' (for example: ca2 , hf2^ , fe4 [fe (cn)6]3, nh4no3, so42 , ch3cooh, cuso4*5h2o). the oxidation state of an atom is the charge of this atom after ionic approximation of its heteronuclear bonds. the oxidation number is synonymous with the. The sum of the oxidation numbers in a polyatomic ion is equal to the charge on that ion. again, work backwards to determine the oxidation number of any non oxygen or non hydrogen atom. to determine the oxidation number of cr in cr 2 o 7 2 : oxygen will be 2 (rule 4), for a total of: 2 × 7 = 14. The sum of all oxidation numbers in a polyatomic ion is equal to the charge on the ion. example: in fe 2 o 3, the oxidation number of fe is 3, and that of o is 2. the sum of all oxidation numbers is: 3 x 2 ( 2) x 3 = 0. the result is expected since fe 2 o 3 is neutral. 4. the oxidation number of an alkali metal in a compound is 1, and the. The oxidation number of a monatomic (composed of one atom) ion is the same as the charge of the ion. for example, the oxidation numbers of k k . , se2 − se 2 −. , and au3 au 3 . are 1 1. , − 2 − 2. , and 3 3. , respectively. the oxidation number of oxygen in most compounds is − 2 − 2.
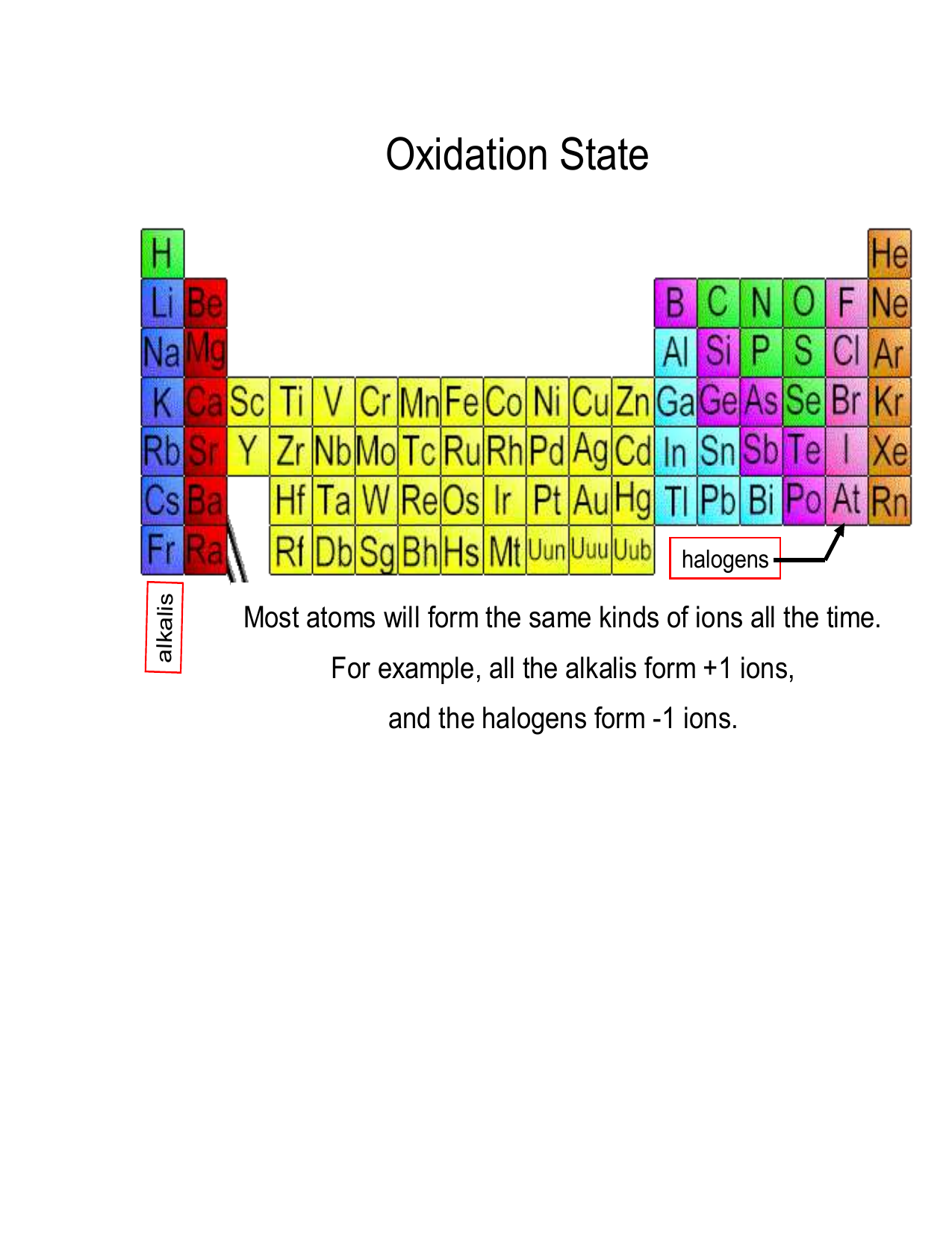
Oxidation Numbers The sum of all oxidation numbers in a polyatomic ion is equal to the charge on the ion. example: in fe 2 o 3, the oxidation number of fe is 3, and that of o is 2. the sum of all oxidation numbers is: 3 x 2 ( 2) x 3 = 0. the result is expected since fe 2 o 3 is neutral. 4. the oxidation number of an alkali metal in a compound is 1, and the. The oxidation number of a monatomic (composed of one atom) ion is the same as the charge of the ion. for example, the oxidation numbers of k k . , se2 − se 2 −. , and au3 au 3 . are 1 1. , − 2 − 2. , and 3 3. , respectively. the oxidation number of oxygen in most compounds is − 2 − 2.

How To Calculate Oxidation Numbers Basic Introduction Youtube
