How To Draw The Lewis Dot Structure For Ch4 Methane Youtube A step by step explanation of how to draw the ch4 lewis dot structure (methane).for the ch4 structure use the periodic table to find the total number of vale. How to draw the lewis dot structure for ch4: methanea step by step explanation of how to draw the ch4 lewis dot structure (methane).for the ch4 structure use.

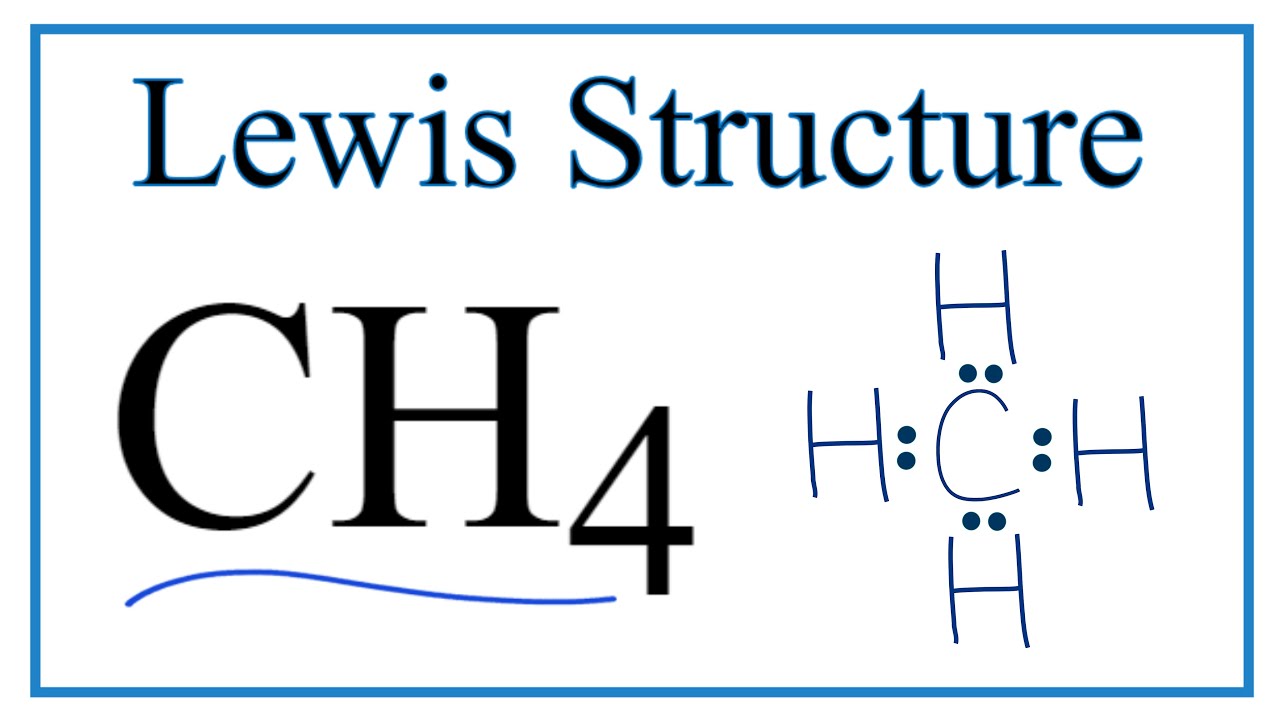
Ch4 Lewis Structure Methane Youtube I quickly take you through how to draw the lewis structure of methane, ch4. i also go over hybridization, shape and bond angle. For ch 4 you have a total of 8 total valence electrons. drawing the lewis structure for ch 4 (named methane) requires only single bonds. it's one of the easier lewis structures to draw. remember that hydrogen atoms always go on the outside of a lewis structure and that they only need two valence electrons for a full outer shell. Step #1: calculate the total number of valence electrons. here, the given molecule is ch4 (methane). in order to draw the lewis structure of ch4, first of all you have to find the total number of valence electrons present in the ch4 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Example 1: draw the lewis structure of methane (ch4). solution: determine the total number of valence electrons: carbon (c) = 4 valence electrons hydrogen (h) = 1 valence electron x 4 = 4 valence electrons total = 4 4 = 8 valence electrons. place the carbon atom in the center and connect it with four hydrogen atoms using single bonds.

How To Draw The Lewis Structure Of Ch4 Methane Youtube Step #1: calculate the total number of valence electrons. here, the given molecule is ch4 (methane). in order to draw the lewis structure of ch4, first of all you have to find the total number of valence electrons present in the ch4 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Example 1: draw the lewis structure of methane (ch4). solution: determine the total number of valence electrons: carbon (c) = 4 valence electrons hydrogen (h) = 1 valence electron x 4 = 4 valence electrons total = 4 4 = 8 valence electrons. place the carbon atom in the center and connect it with four hydrogen atoms using single bonds. Dr. b. explains how to draw the lewis dot structure for ch4 (methane). the ch 4 lewis structure is one of the most frequently tested lewis structures. note that hydrogen atoms always go on the outside of a lewis dot structure. this is because they can share a maximum of two electrons. once they have two valence electrons their outer shell is. In the above structure, there are no charges on carbon and hydrogen atoms. therefore, we can think, we have obtained the lewis structure of ch 4. methane lewis structure contains four c h bonds. there are no lone pairs in the valence shells of carbon atom. carbon atom is the center atom and it is very easy to draw ch4 lewis structure.
