
How To Draw The Lewis Structure For Ionic Compounds Nacl Mgf2 This short flash animation looks briefly at how the ionic bond forms in magnesium fluoride (mgf2).this video shows you how to draw the lewis structure for io. This chemistry video explains how to draw the lewis structures of ionic compounds. examples include nacl, mgf2, k2o, and al2o3.how to draw lewis structures:.

How To Draw The Lewis Structures Of Ionic Compounds Youtube A step by step explanation of how to draw the nacl lewis dot structure (sodium chloride).for nacl we have an ionic compound and we need to take that into acc. Here, the valence electrons in fluorine are 9. now, let’s begin with drawing the lewis structure of magnesium fluoride (mgf2): step 1: search for the maximum available valence electrons in one magnesium fluoride molecule: it is sixteen as two are with magnesium atom and fourteen are with two fluorine atoms. step 2: look for valence electrons. Crystal structure and unit cell. nacl has a cubic crystal system and a face centered cubic crystalline structure. nacl has four cations and four anions in a face centered cubic unit cell. since there are 4 atoms or ions present in each unit cell of a face centred cubic structure, therefore, the number of nacl units in a unit cell of nacl is four. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. 10.3: lewis structures of ionic compounds electrons transferred is shared under a license and was authored, remixed, and or curated by libretexts. the tendency to form species that have eight electrons in the valence shell is called the octet rule.

How To Draw The Lewis Structure For Ionic Compounds Nacl Mgf2 вђ Otosection Crystal structure and unit cell. nacl has a cubic crystal system and a face centered cubic crystalline structure. nacl has four cations and four anions in a face centered cubic unit cell. since there are 4 atoms or ions present in each unit cell of a face centred cubic structure, therefore, the number of nacl units in a unit cell of nacl is four. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. 10.3: lewis structures of ionic compounds electrons transferred is shared under a license and was authored, remixed, and or curated by libretexts. the tendency to form species that have eight electrons in the valence shell is called the octet rule. For example, copper can form ions with a 1 or 2 charge, and iron can form ions with a 2 or 3 charge. figure 4.4.1 4.4. 1: some elements exhibit a regular pattern of ionic charge when they form ions. group one of the periodic table contains l i superscript plus sign in period 2, n a superscript plus sign in period 3, k superscript plus sign. Step #1: calculate the total number of valence electrons. here, the given molecule is mgf2. in order to draw the lewis structure of mgf2, first of all you have to find the total number of valence electrons present in the mgf2 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom).
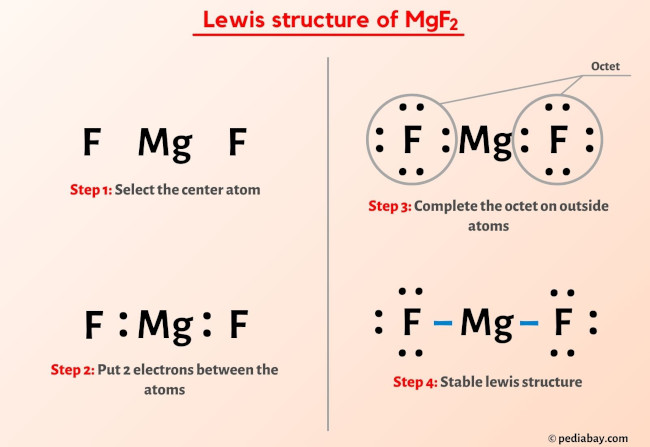
Mgf2 Lewis Structure In 6 Steps With Images For example, copper can form ions with a 1 or 2 charge, and iron can form ions with a 2 or 3 charge. figure 4.4.1 4.4. 1: some elements exhibit a regular pattern of ionic charge when they form ions. group one of the periodic table contains l i superscript plus sign in period 2, n a superscript plus sign in period 3, k superscript plus sign. Step #1: calculate the total number of valence electrons. here, the given molecule is mgf2. in order to draw the lewis structure of mgf2, first of all you have to find the total number of valence electrons present in the mgf2 molecule. (valence electrons are the number of electrons present in the outermost shell of an atom).

Ionic Bond Represented By Lewis Dot Structure Nacl And Mgf2 Science
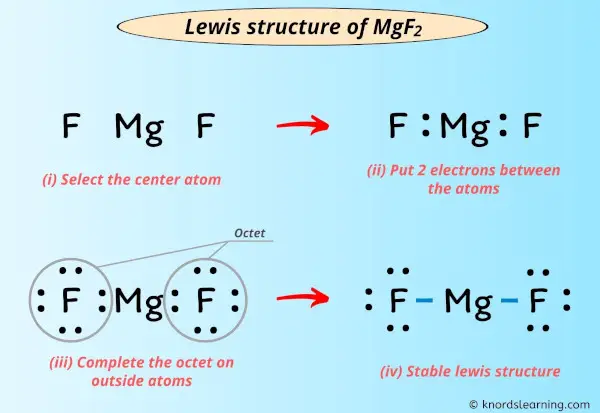
Lewis Structure Of Mgf2 With 6 Simple Steps To Draw
