
How To Draw Lewis Structures Youtube A video tutorial for how to draw lewis structures in five steps. more lewis structures help at breslyn.org (and get all my free chem video guid. This chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as cl2, o2, of2, ch4, nh3, h2o, c2h2, and n2h4 .
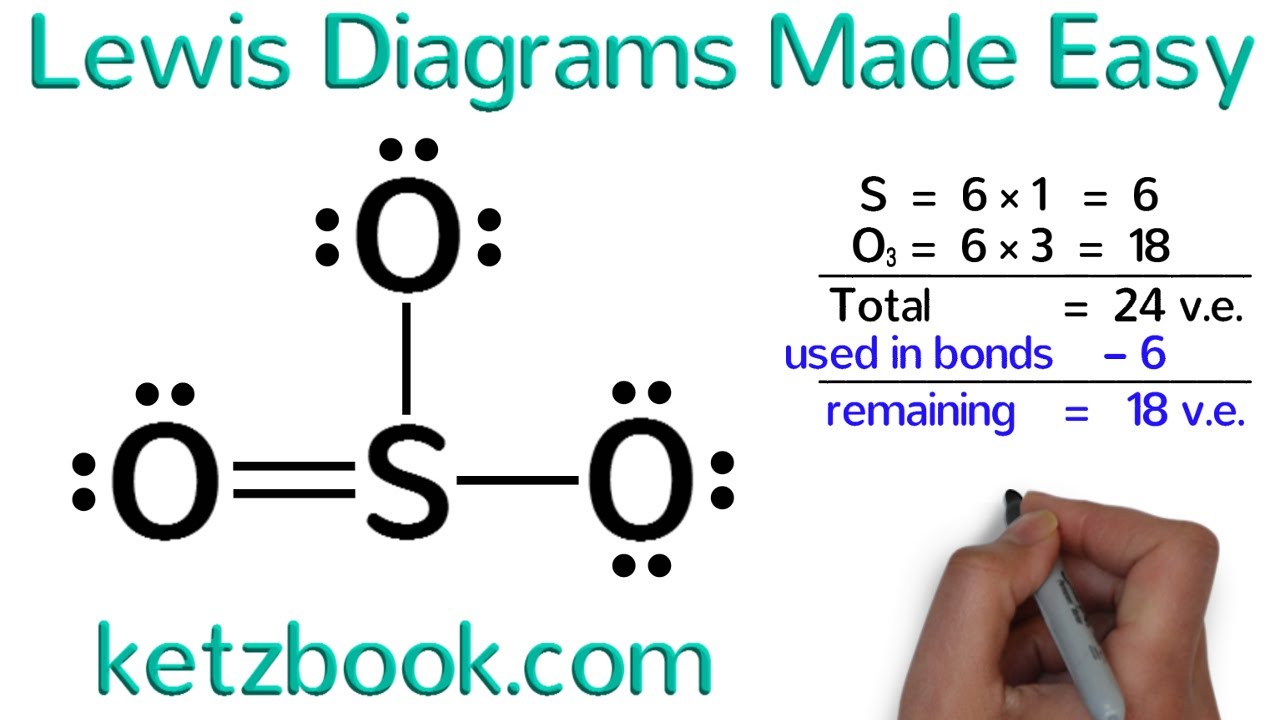
Lewis Diagrams Made Easy How To Draw Lewis Dot Structures Youtube This organic chemistry video tutorial explains how to draw lewis structures using a simple method. organic chemistry basic introduction: https:. A lewis structure includes lines for covalent chemical bonds and dots for valence electrons or lone electron pairs. different ways to draw lewis structures. there is more than one “right” way to draw a lewis structure. if you are drawing the structures for a chemistry class, be sure to know what your instructor expects. Examples for drawing lewis structures for covalent bonds . here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. reference the “how to draw a lewis dot structure” for a step by step guide. see the following lewis dot structure diagrams for a few covalent compounds. example 1. Lewis structures (also known as lewis dot diagrams, electron dot diagrams,"lewis dot formula" lewis dot structures, and electron dot structures) are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. a lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. the lewis structure.
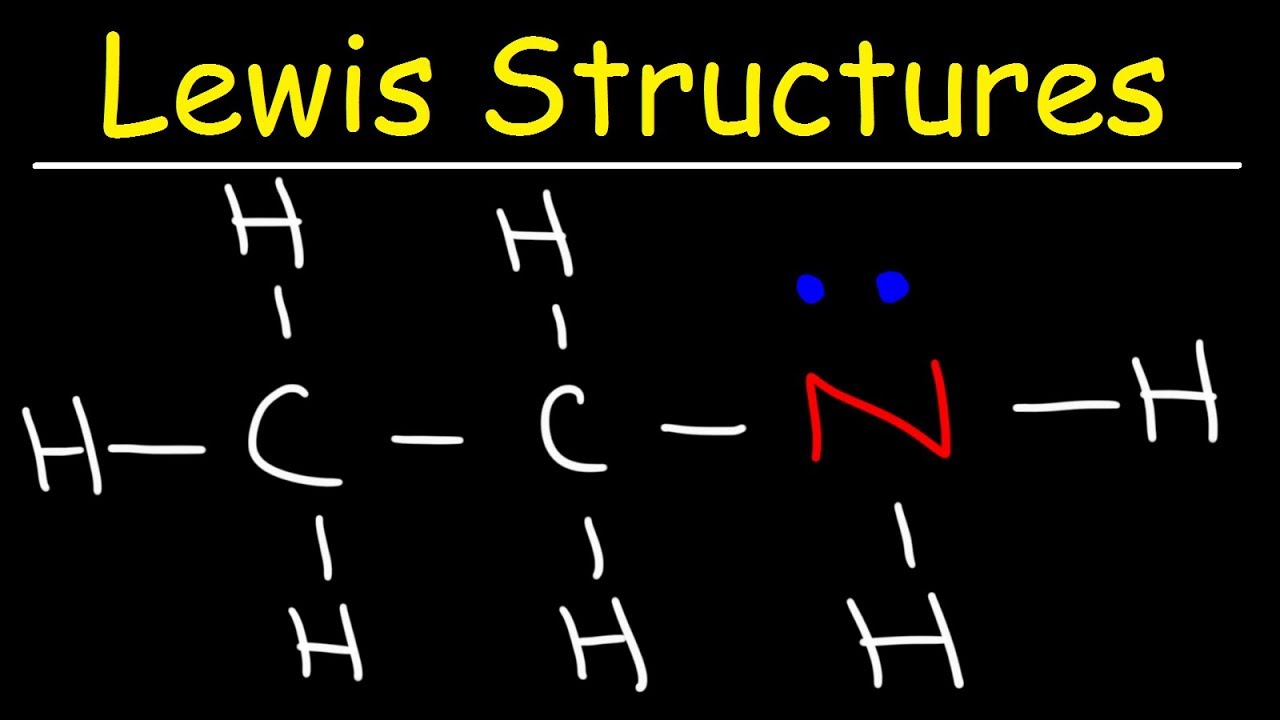
Organic Chemistry How To Draw Lewis Structures Youtube Examples for drawing lewis structures for covalent bonds . here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. reference the “how to draw a lewis dot structure” for a step by step guide. see the following lewis dot structure diagrams for a few covalent compounds. example 1. Lewis structures (also known as lewis dot diagrams, electron dot diagrams,"lewis dot formula" lewis dot structures, and electron dot structures) are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. a lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. the lewis structure. Step 3: determine the number of bonds in the molecule. covalent bonds are formed when one electron from each atom forms an electron pair. step 2 tells how many electrons are needed and step 1 is how many electrons you have. subtracting the number in step 1 from the number in step 2 gives you the number of electrons needed to complete the octets. Step 1: figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. when drawing the structure of an ion, be sure to add subtract electrons to account for the charge. step 2: connect the atoms to each other with single bonds to form a “skeleton structure.”.

Quick Easy 5 Steps To Drawing Lewis Structures With Examples Step 3: determine the number of bonds in the molecule. covalent bonds are formed when one electron from each atom forms an electron pair. step 2 tells how many electrons are needed and step 1 is how many electrons you have. subtracting the number in step 1 from the number in step 2 gives you the number of electrons needed to complete the octets. Step 1: figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. when drawing the structure of an ion, be sure to add subtract electrons to account for the charge. step 2: connect the atoms to each other with single bonds to form a “skeleton structure.”.

How To Draw The Lewis Structures Of Ionic Compounds Youtube

Beginner S Guide To Drawing Lewis Structures Youtube
