
How To Draw The Lewis Structures Of Ionic Compounds Youtube When drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent molecular compounds. Shows how to draw lewis dot structures for ionic compounds. you can see a listing of all my videos at my website, stepbystepscience.

How To Draw Lewis Dot Structures For Ionic Compounds Bonds This chemistry video explains how to draw the lewis structures of ionic compounds. examples include nacl, mgf2, k2o, and al2o3.how to draw lewis structures:. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. 10.3: lewis structures of ionic compounds electrons transferred is shared under a license and was authored, remixed, and or curated by libretexts. the tendency to form species that have eight electrons in the valence shell is called the octet rule. A lewis structure includes lines for covalent chemical bonds and dots for valence electrons or lone electron pairs. different ways to draw lewis structures. there is more than one “right” way to draw a lewis structure. if you are drawing the structures for a chemistry class, be sure to know what your instructor expects. For anions add a number of electrons equal to the negative charge. for cations subtract a number of electrons equal to the positive charge. 4. place one electron pair between each pair of adjacent atoms (as determined from the framework found in step 2) to form a single bond. 5.
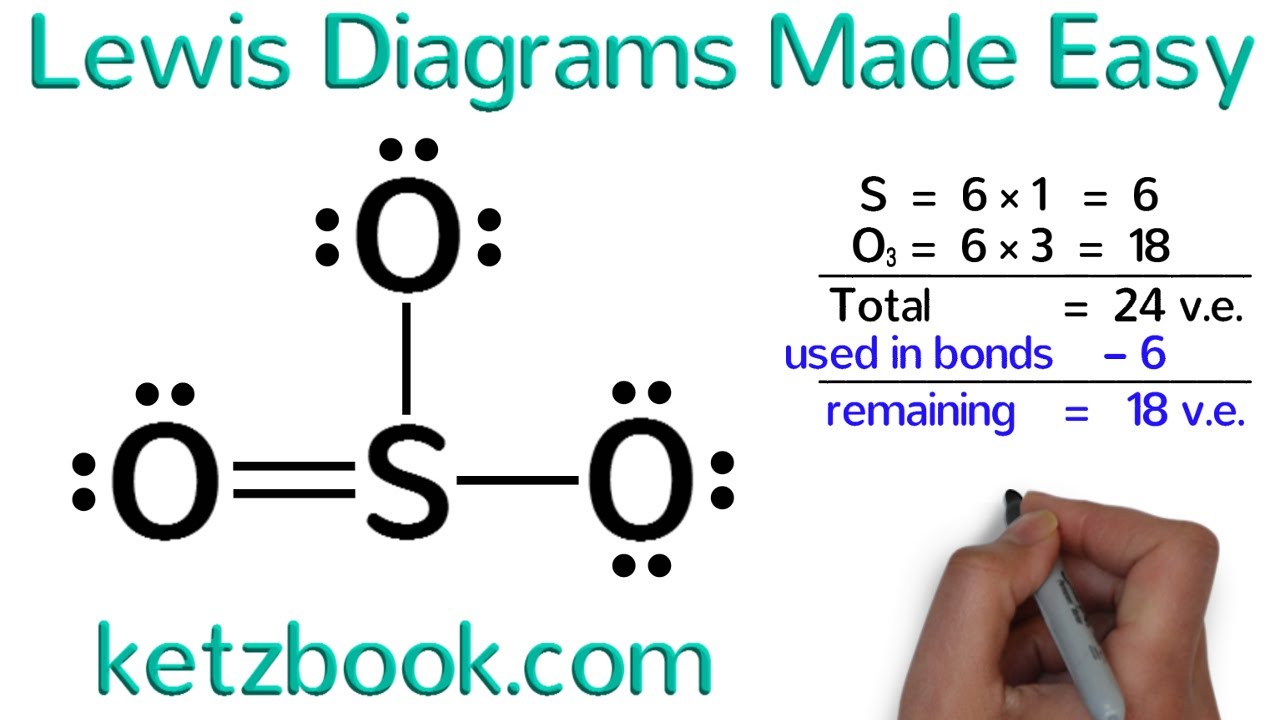
Lewis Diagrams Made Easy How To Draw Lewis Dot Structures Youtubeођ A lewis structure includes lines for covalent chemical bonds and dots for valence electrons or lone electron pairs. different ways to draw lewis structures. there is more than one “right” way to draw a lewis structure. if you are drawing the structures for a chemistry class, be sure to know what your instructor expects. For anions add a number of electrons equal to the negative charge. for cations subtract a number of electrons equal to the positive charge. 4. place one electron pair between each pair of adjacent atoms (as determined from the framework found in step 2) to form a single bond. 5. Examples for drawing lewis structures for covalent bonds . here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. reference the “how to draw a lewis dot structure” for a step by step guide. see the following lewis dot structure diagrams for a few covalent compounds. example 1. Step 1: figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. when drawing the structure of an ion, be sure to add subtract electrons to account for the charge. step 2: connect the atoms to each other with single bonds to form a “skeleton structure.”.

How To Draw Lewis Structures Youtube Examples for drawing lewis structures for covalent bonds . here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. reference the “how to draw a lewis dot structure” for a step by step guide. see the following lewis dot structure diagrams for a few covalent compounds. example 1. Step 1: figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. when drawing the structure of an ion, be sure to add subtract electrons to account for the charge. step 2: connect the atoms to each other with single bonds to form a “skeleton structure.”.

Formation Of Ionic Compounds Using Dot Structures Youtube
