
How To Draw Hbr Lewis Structure Science Education And Vrogue Co The lewis structure (lewis dot diagram) for co.1. count electrons2. put least electronegative atom in centre3. put one electron pair in each bond4. fill oute. A step by step explanation of how to draw the co lewis dot structure (carbon monoxide ).for the co structure use the periodic table to find the total number.
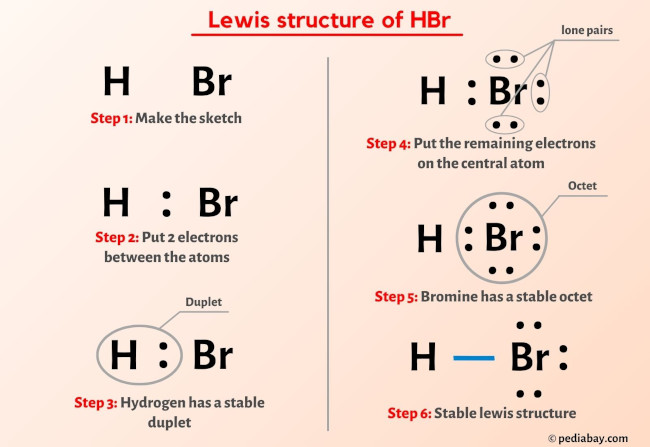
How To Draw Hbr Lewis Structure Science Education And Vrogue Co Step 1: figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. when drawing the structure of an ion, be sure to add subtract electrons to account for the charge. step 2: connect the atoms to each other with single bonds to form a “skeleton structure.”. Step 1: determine the total number of valence electrons. the first step in drawing the lewis structure of carbon monoxide is to determine the total number of valence electrons present in the molecule. valence electrons are the electrons in the outermost energy level of an atom that participate in chemical bonding. A lewis structure includes lines for covalent chemical bonds and dots for valence electrons or lone electron pairs. different ways to draw lewis structures. there is more than one “right” way to draw a lewis structure. if you are drawing the structures for a chemistry class, be sure to know what your instructor expects. A step by step explanation of how to draw the hbr lewis dot structure (hydrogen bromide).for the hbr structure use the periodic table to find the total numbe.

How To Draw Hbr Lewis Structure Science Education And Vrogue Co A lewis structure includes lines for covalent chemical bonds and dots for valence electrons or lone electron pairs. different ways to draw lewis structures. there is more than one “right” way to draw a lewis structure. if you are drawing the structures for a chemistry class, be sure to know what your instructor expects. A step by step explanation of how to draw the hbr lewis dot structure (hydrogen bromide).for the hbr structure use the periodic table to find the total numbe. Step #1: calculate the total number of valence electrons. here, the given molecule is co (carbon monoxide). in order to draw the lewis structure of co, first of all you have to find the total number of valence electrons present in the co molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). How to draw the lewis structure for co. drawing the co lewis structure involves several steps: 1. determine the total number of valence electrons in co. to determine the total number of valence electrons in co, you need to add up the valence electrons of each atom in the molecule. carbon (c) has 4 valence electrons, and oxygen (o) has 6 valence.
How To Draw Hbr Lewis Structure Science Education And Vrogue Co Step #1: calculate the total number of valence electrons. here, the given molecule is co (carbon monoxide). in order to draw the lewis structure of co, first of all you have to find the total number of valence electrons present in the co molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). How to draw the lewis structure for co. drawing the co lewis structure involves several steps: 1. determine the total number of valence electrons in co. to determine the total number of valence electrons in co, you need to add up the valence electrons of each atom in the molecule. carbon (c) has 4 valence electrons, and oxygen (o) has 6 valence.
How To Draw Hbr Lewis Structure Science Education And Vrogue Co
