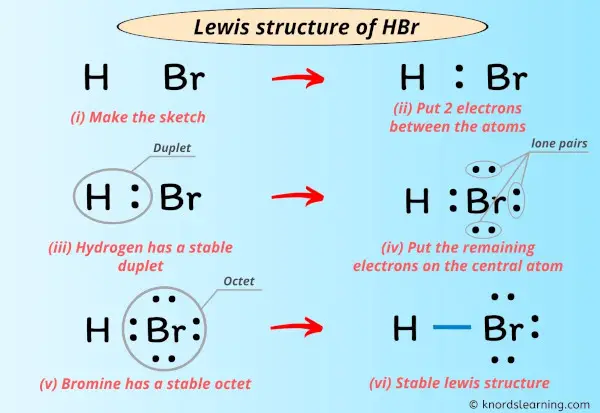
Lewis Structure Of Hbr With 6 Simple Steps To Draw Hbr lewis structure, molecular geometry, hybridization, and mo diagram. hydrogen bromide, hbr is a hydrogen halide compound owing to the fact that bromine belongs to the halogen family. quite a corrosive and dangerous chemical, it can be useful too in a lot of ways. it is used to prepare a variety of organic and inorganic bromine compounds. A step by step explanation of how to draw the hbr lewis dot structure (hydrogen bromide).for the hbr structure use the periodic table to find the total numbe.
Lewis Structure Of Hbr With 6 Simple Steps To Draw The hbr lewis structure represents the arrangement of atoms and bonding electrons in a molecule of hydrogen bromide (hbr). it showcases the connectivity between hydrogen (h) and bromine (br) atoms, giving us a visual representation of their covalent bond. atomic information: hydrogen (h): 1 proton, 1 electron bromine (br): 35 protons, 35 electrons drawing the. Modelset: havesymmetry:false haveunitcells:false havefractionalcoord:false. 1 model in this collection. use getproperty "modelinfo" or getproperty "auxiliaryinfo" to inspect them. modelset: not autobonding; use forceautobond=true to force automatic bond creation. Hydrogen bromide (hbr) lewis structure. lewis structure of hydrogen bromide (hbr) contains only one h br bond. there are no charges on atoms in hbr lewis structure because hbr is a neutral molecule. there is three lone pairs on bromine atom in hbr molecule. hbr is a very easy lewis structure to draw due to its simplicity. Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom).

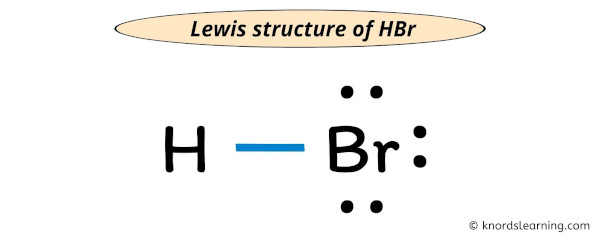
Hbr Lewis Structure Molecular Geometry Hybridization And Mo Diagram Hydrogen bromide (hbr) lewis structure. lewis structure of hydrogen bromide (hbr) contains only one h br bond. there are no charges on atoms in hbr lewis structure because hbr is a neutral molecule. there is three lone pairs on bromine atom in hbr molecule. hbr is a very easy lewis structure to draw due to its simplicity. Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). In the above structure, you can see that the central atom (bromine) forms an octet. and the outside atom (hydrogen) also forms a duet. hence, the octet rule and duet rule are satisfied. therefore, this structure is the stable lewis structure of hbr. next: n 2 h 4 lewis structure your feedback matters. visit our contact page. This structure is also available as a 2d mol file or as a computed 3d sd file the 3d structure may be viewed using java or javascript . other names: hydrobromic acid; anhydrous hydrobromic acid; hbr; hydrogen bromide, anhydrous ; acide bromhydrique; acido bromidrico; bromowodor; bromwasserstoff; broomwaterstof; un 1048; un 1788; hydrogen monobromide.

Hbr Molecular Geometry Bond Angles And Electron Geometry Youtube In the above structure, you can see that the central atom (bromine) forms an octet. and the outside atom (hydrogen) also forms a duet. hence, the octet rule and duet rule are satisfied. therefore, this structure is the stable lewis structure of hbr. next: n 2 h 4 lewis structure your feedback matters. visit our contact page. This structure is also available as a 2d mol file or as a computed 3d sd file the 3d structure may be viewed using java or javascript . other names: hydrobromic acid; anhydrous hydrobromic acid; hbr; hydrogen bromide, anhydrous ; acide bromhydrique; acido bromidrico; bromowodor; bromwasserstoff; broomwaterstof; un 1048; un 1788; hydrogen monobromide.
