
Fe C Phase Diagram And Microstructures Download Scientific Diagram Summary. phase diagrams are useful tools to determine: the number and types of phases, the wt% of each phase, and the composition of each phase. for a given t and composition of the system. alloying to produce a solid solution usually. increases the tensile strength (ts) decreases the ductility. binary eutectics and binary eutectoids. The fe c phase diagram is primarily composed of two elements: iron (fe) and carbon (c). it showcases the different phases that occur when different concentrations of carbon are added to iron. the diagram consists of a temperature axis and a carbon concentration axis. on the left side of the diagram, we have pure iron, known as the α iron phase.

Fe Phase Diagram The diagram describes the suitable conditions for two or more phases to exist in equilibrium. for example, the water phase diagram describes a point (triple point) where water can coexist in three different phases at the same time. this happens at just above the freezing temperature (0.01°c) and 0.006 atm. using the diagrams. there are four. The fe c phase diagram elucidates the relationship between carbon content, temperature, and the resulting microstructure, enabling engineers to predict and control the mechanical, thermal, and chemical properties of steels. the phase diagram reveals several significant phases, including ferrite, austenite, cementite, and various combinations of. The fe c diagram, also known as the iron carbon phase diagram, is a critical tool in understanding the behavior and properties of iron and steel. it provides valuable information about the phases and transformations that occur during the cooling and heating of iron carbon alloys. one of the key features of the fe c diagram is the presence of. A metastable iron carbon phase diagram is used to emphasize the fact that the iron carbon relationship is metastable due to the slow cooling rates of iron alloys. in the term “fe c phase diagram,” fe c is shorthand for iron carbon. the diagram is also known as a “steel phase diagram” since iron alloys are steel.
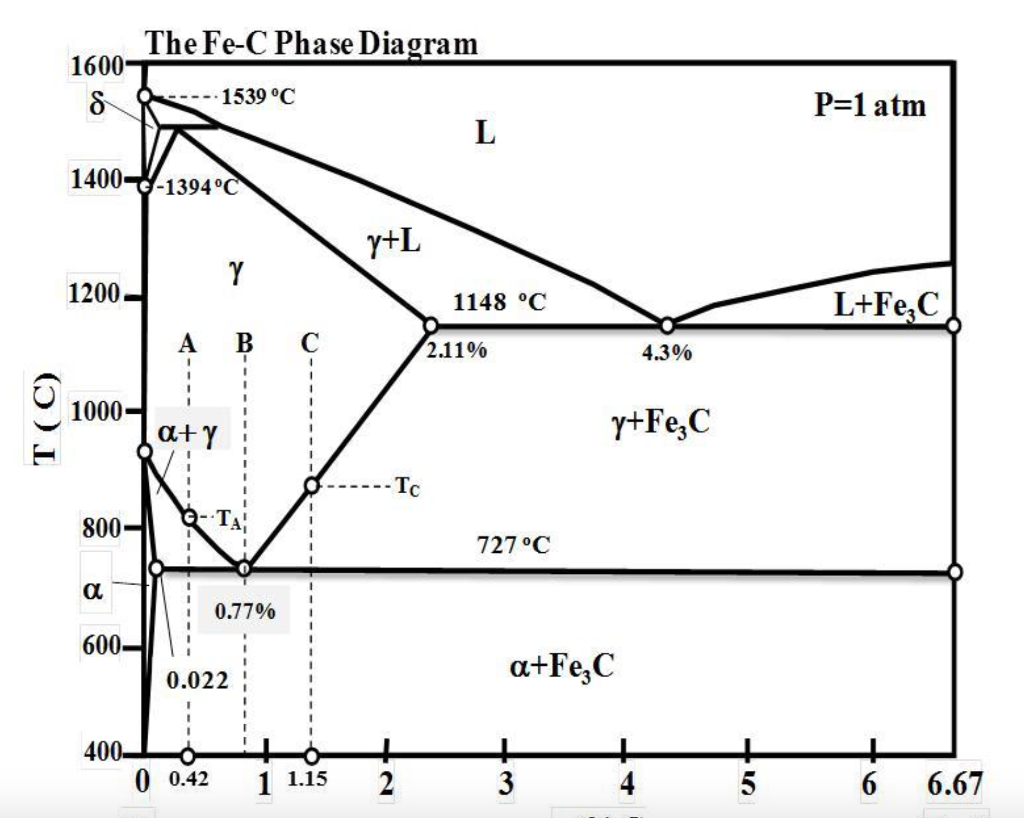
Solved By Using The Iron Carbon Fe C Phase Diagram Prov Chegg The fe c diagram, also known as the iron carbon phase diagram, is a critical tool in understanding the behavior and properties of iron and steel. it provides valuable information about the phases and transformations that occur during the cooling and heating of iron carbon alloys. one of the key features of the fe c diagram is the presence of. A metastable iron carbon phase diagram is used to emphasize the fact that the iron carbon relationship is metastable due to the slow cooling rates of iron alloys. in the term “fe c phase diagram,” fe c is shorthand for iron carbon. the diagram is also known as a “steel phase diagram” since iron alloys are steel. Steel production: the fe c phase diagram allows steelmakers to determine the appropriate composition and heat treatment processes required to produce desired steel properties. by understanding the phase transformations that occur during cooling and heating, steel manufacturers can manipulate the microstructure and properties of the final product. Summary. phase diagrams are useful tools to determine: the number and types of phases, the wt% of each phase, and the composition of each phase. for a given t and composition of the system. alloying to produce a solid solution usually. increases the tensile strength (ts) decreases the ductility.

Solved Using The Fe C Phase Diagram And Diagram For A Chegg Steel production: the fe c phase diagram allows steelmakers to determine the appropriate composition and heat treatment processes required to produce desired steel properties. by understanding the phase transformations that occur during cooling and heating, steel manufacturers can manipulate the microstructure and properties of the final product. Summary. phase diagrams are useful tools to determine: the number and types of phases, the wt% of each phase, and the composition of each phase. for a given t and composition of the system. alloying to produce a solid solution usually. increases the tensile strength (ts) decreases the ductility.
