
Electron Dot Structure Of Ethyne C2h2 For c 2 h 2 you have a total of 10 valence electrons to work with. in drawing the lewis structure for c 2 h 2 (also called ethyne) you'll find that you don't have enough valence electrons available to satisfy the octet for each element (if you use only single bonds). the solution is to share three pairs of valence electrons and form a triple. A step by step explanation of how to draw the c2h2 lewis dot structure (ethyne or acetylene).for the c2h2 structure use the periodic table to find the total.

Electron Dot Structure Of Ethyne C2h2 The lewis structure for c2h2, also known as ethyne or acetylene, is a diagram that shows the arrangement of valence electrons and the bonding between atoms in a molecule. this structure is essential in understanding the properties and behavior of c2h2 in chemical reactions. c2h2 is a hydrocarbon compound made up of two carbon atoms and two. A video explanation of how to draw the lewis dot structure for acetylene or ethyne, along with information about the compound including formal charges, polar. A step by step explanation of how to draw the c2h2 lewis dot structure (acetylene (ethyne)).for the c2h2 structure use the periodic table to find the total n. C2h2 lewis structure. lewis structure of any molecule helps to know the arrangement of all atoms, their valence electrons, and the bond formation in the molecule. the electrons that participate in forming bonds are called bonding pairs of electrons while the ones that do not take part in any bond formation are called lone pairs or non bonding pairs of electrons.

C2h2 Lewis Structure Ethyne Or Acetylene Ethyne Lewis Dot A step by step explanation of how to draw the c2h2 lewis dot structure (acetylene (ethyne)).for the c2h2 structure use the periodic table to find the total n. C2h2 lewis structure. lewis structure of any molecule helps to know the arrangement of all atoms, their valence electrons, and the bond formation in the molecule. the electrons that participate in forming bonds are called bonding pairs of electrons while the ones that do not take part in any bond formation are called lone pairs or non bonding pairs of electrons. Ethylene (commonly knows as ethene), ch 2 ch 2, is the simplest molecule which contains a carbon carbon double bond. the lewis structure of ethylene indicates that there are one carbon carbon double bond and four carbon hydrogen single bonds. experimentally, the four carbon hydrogen bonds in the ethylene molecule have been shown to be identical. It was recognized nearly 150 years ago, however, that carbon atoms can also form double bonds by sharing two electron pairs between atoms or triple bonds by sharing three electron pairs. ethylene, for instance, has the structure h 2 c=c h 2 h 2 c=c h 2 and contains a carbon–carbon double bond, while acetylene has the structure hc≡ch hc≡ch.
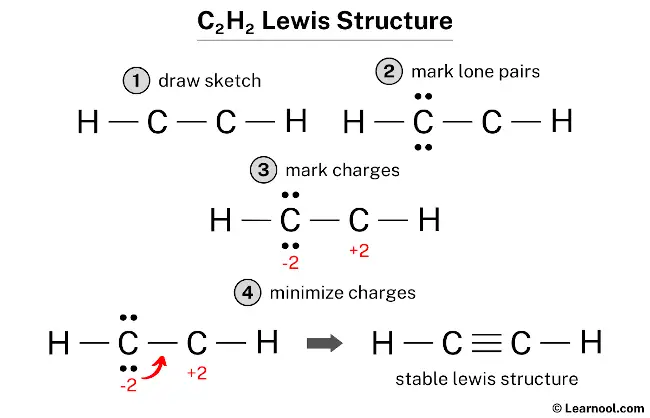
C2h2 Lewis Structure Learnool Ethylene (commonly knows as ethene), ch 2 ch 2, is the simplest molecule which contains a carbon carbon double bond. the lewis structure of ethylene indicates that there are one carbon carbon double bond and four carbon hydrogen single bonds. experimentally, the four carbon hydrogen bonds in the ethylene molecule have been shown to be identical. It was recognized nearly 150 years ago, however, that carbon atoms can also form double bonds by sharing two electron pairs between atoms or triple bonds by sharing three electron pairs. ethylene, for instance, has the structure h 2 c=c h 2 h 2 c=c h 2 and contains a carbon–carbon double bond, while acetylene has the structure hc≡ch hc≡ch.
