
Drug Delivery Devices Throughout The Drug Development Cycle Eitan Medical Share. step 1 discovery and development. discovery and development research for a new drug begins in the laboratory. more information. step 2 preclinical research. preclinical research drugs. The drug approval process takes place within a structured framework that includes: analysis of the target condition and available treatments —fda reviewers analyze the condition or illness for.

2021 Fda Drug Approval Process Infographic Conquer The Journey Informed Overview how drugs are developed: this web page provides an example on how a drug sponsor can work with fda's regulations and guidance information to bring a new drug to market, from clinical. Fda drug approval process. The drug development and approval process. the process of getting a drug to market, from first testing to final fda approval, is summarized in figure 1 and described at greater length below. drug companies continuously analyze thousands of compounds, seeking ones of therapeutic value. during the six to seven years of preclinical testing, the. The fda serves as a regulatory agency ensuring safety, efficacy, and quality are met in the approval of drugs, vaccines, biologics, and testing products. understanding the terms of development can help providers with many new products being developed. keywords: drug development, eua, fda, regulation. over the past year, the severe acute.
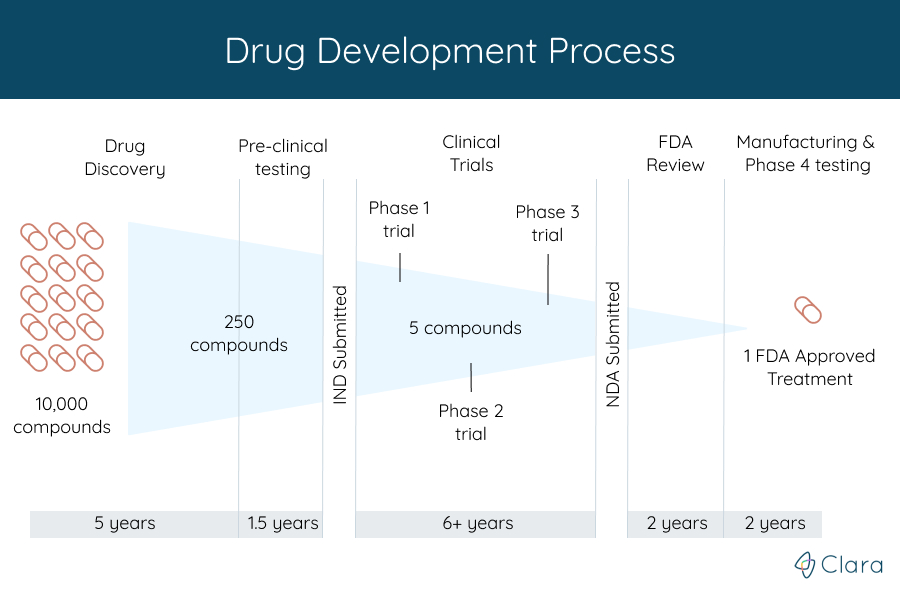
What Is A Clinical Trial Find Register For Fda Clinical Trials The drug development and approval process. the process of getting a drug to market, from first testing to final fda approval, is summarized in figure 1 and described at greater length below. drug companies continuously analyze thousands of compounds, seeking ones of therapeutic value. during the six to seven years of preclinical testing, the. The fda serves as a regulatory agency ensuring safety, efficacy, and quality are met in the approval of drugs, vaccines, biologics, and testing products. understanding the terms of development can help providers with many new products being developed. keywords: drug development, eua, fda, regulation. over the past year, the severe acute. Drug development is the process of bringing a novel drug from “bench to bedside”. it can take 10 to 15 years for a drug to be designed, developed and approved for use in patients (fig 1). in some circumstances, the drug development and approval process can be expedited – for example, if the drug is the first available treatment for a. For researchers involved in the clinical development and testing of putative drugs and devices, the process of fda approval can be daunting and difficult to navigate. this first part of a 2 part series is intended to provide an overview of the steps in bringing a drug through the process of clinical trials and fda approval.

Drug Development Strategy For Us Fda Prorelix Research Drug development is the process of bringing a novel drug from “bench to bedside”. it can take 10 to 15 years for a drug to be designed, developed and approved for use in patients (fig 1). in some circumstances, the drug development and approval process can be expedited – for example, if the drug is the first available treatment for a. For researchers involved in the clinical development and testing of putative drugs and devices, the process of fda approval can be daunting and difficult to navigate. this first part of a 2 part series is intended to provide an overview of the steps in bringing a drug through the process of clinical trials and fda approval.
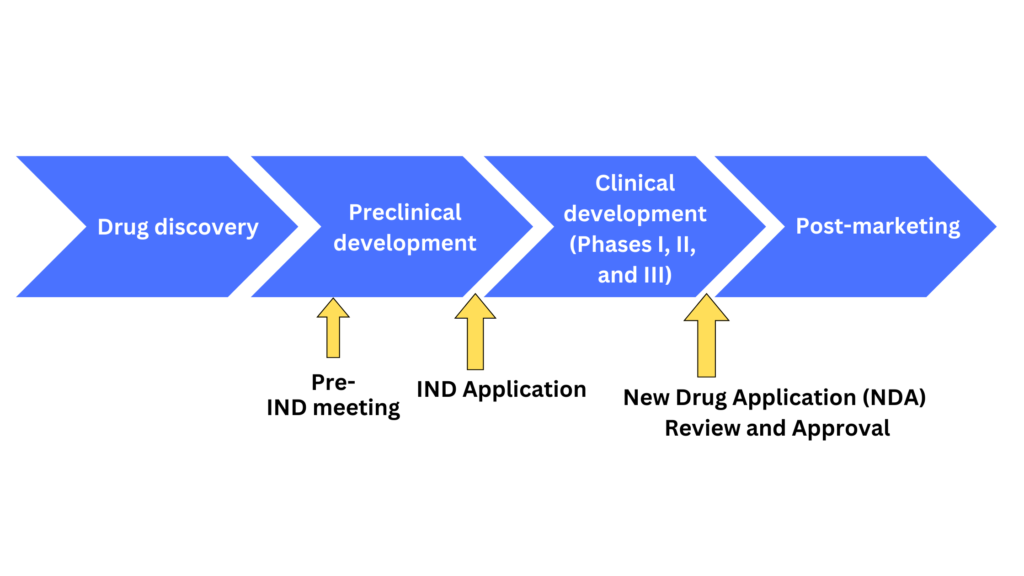
Ind Data Requirements And Us Fda Submission Process
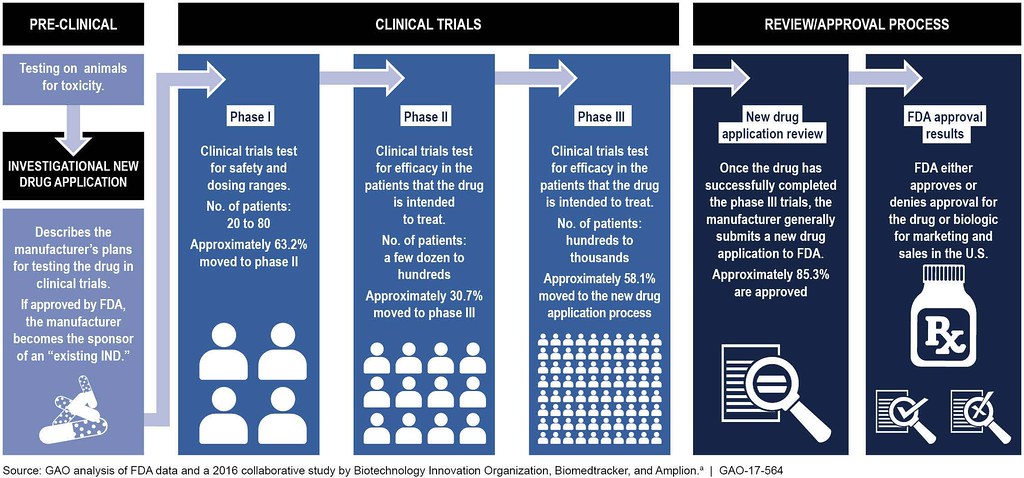
6 2 The Drug Development Process Biology Libretexts
