
Hydrogen Bromide Lewis Dot Structure A step by step explanation of how to draw the hbr lewis dot structure (hydrogen bromide).for the hbr structure use the periodic table to find the total numbe. The hbr lewis structure represents the arrangement of atoms and bonding electrons in a molecule of hydrogen bromide (hbr). it showcases the connectivity between hydrogen (h) and bromine (br) atoms, giving us a visual representation of their covalent bond. atomic information: hydrogen (h): 1 proton, 1 electron bromine (br): 35 protons, 35 electrons drawing the.

Draw The Lewis Structure Of Hbr Hydrogen Bromide 41 Off Lewis structure of hydrogen bromide (hbr) contains only one h br bond. there are no charges on atoms in hbr lewis structure because hbr is a neutral molecule. there is three lone pairs on bromine atom in hbr molecule. hbr is a very easy lewis structure to draw due to its simplicity. Hbr lewis structure, molecular geometry, hybridization, and mo diagram. hydrogen bromide, hbr is a hydrogen halide compound owing to the fact that bromine belongs to the halogen family. quite a corrosive and dangerous chemical, it can be useful too in a lot of ways. it is used to prepare a variety of organic and inorganic bromine compounds. Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Here’s how you can easily draw the hbr lewis structure step by step: #1 draw a rough skeleton structure. #2 mention lone pairs on the atoms. #3 if needed, mention formal charges on the atoms. now, let’s take a closer look at each step mentioned above.

Draw The Lewis Structure Of Hbr Hydrogen Bromide Youtube Step #1: calculate the total number of valence electrons. here, the given molecule is hbr (hydrogen bromide). in order to draw the lewis structure of hbr, first of all you have to find the total number of valence electrons present in the hbr molecule. (valence electrons are the number of electrons present in the outermost shell of an atom). Here’s how you can easily draw the hbr lewis structure step by step: #1 draw a rough skeleton structure. #2 mention lone pairs on the atoms. #3 if needed, mention formal charges on the atoms. now, let’s take a closer look at each step mentioned above. Drawing the lewis structure for hbr. hbr is very similar to hf and hcl. hydrogen has 1 valence electron and br (in group 7 with f and cl) has 7 valence electrons. with the lewis structure for hbr remember that hydrogen only needs 2 valence electrons to have a full outer shell. be sure that you don't use more than the 8 valence electrons available. Figure 11.3m: lewis structures of the formation of hydrogen bromide, hbr, hydrogen sulfide, h 2 s, and nitrogen, n 2 (credit: chemistry (openstax), cc by 4.0). for more complicated molecules and molecular ions, it is helpful to follow the step by step procedure outlined here: determine the total number of valence (outer shell) electrons.
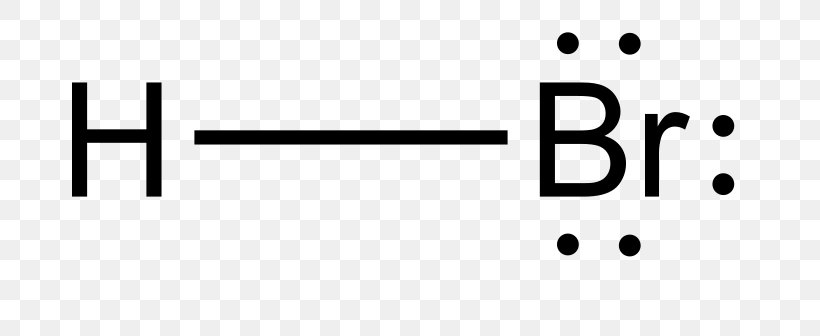
Draw The Lewis Structure Of Hbr Hydrogen Bromide Drawing the lewis structure for hbr. hbr is very similar to hf and hcl. hydrogen has 1 valence electron and br (in group 7 with f and cl) has 7 valence electrons. with the lewis structure for hbr remember that hydrogen only needs 2 valence electrons to have a full outer shell. be sure that you don't use more than the 8 valence electrons available. Figure 11.3m: lewis structures of the formation of hydrogen bromide, hbr, hydrogen sulfide, h 2 s, and nitrogen, n 2 (credit: chemistry (openstax), cc by 4.0). for more complicated molecules and molecular ions, it is helpful to follow the step by step procedure outlined here: determine the total number of valence (outer shell) electrons.

Solved Draw The Lewis Structure For A Hydrogen Bromide Hbr Molec
