
Difference Between Free Radical And Ion Compare The Difference Free radicals and ions can both act as intermediates in various reaction mechanisms, facilitating the formation of new compounds. however, the key difference lies in their electronic configuration. free radicals possess an unpaired electron, making them highly reactive, while ions have a complete electron configuration, rendering them more stable. Key differences. free radicals are chemical species with one or more unpaired electrons, making them highly reactive and unstable. ions, in contrast, are atoms or molecules that have gained or lost electrons, resulting in a net positive or negative charge. the reactivity of free radicals stems from their unpaired electrons seeking stability.
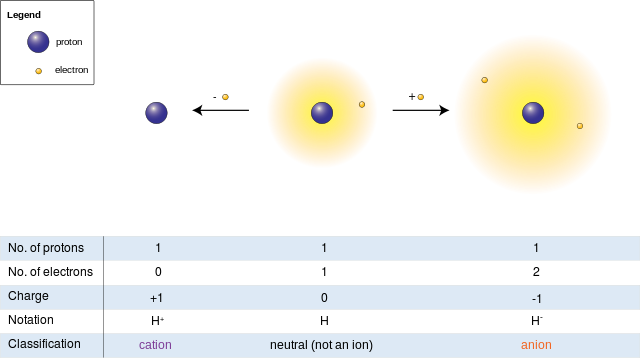
Difference Between Free Radical And Ion Compare The Difference They are formed through the process of ionization and are often involved in chemical reactions. on the other hand, radicals are neutral species that have an unpaired electron in their outermost shell. they are highly reactive and can initiate chain reactions in various chemical processes. while ions are more stable due to their charge, radicals. Also, ions in nature tend to be compensated by ions with opposite charges, whereas radicals are very reactive and thus short lived. answer 3: an ion has a non zero electric charge. a radical has an atom with unfilled electron shells and so is very reactive, but is electrically neutral. answer 4: an ion carries a charge, that means that the. Free radical: highly reactive due to the presence of an unpaired electron, seeking to pair it, which can lead to chain reactions. ion: its reactivity depends on the charge and the desire to return to a neutral state. ions can be relatively more stable than free radicals. One of the main differences between ions and free radicals is their stability. ions are generally stable and do not readily react with other substances, while free radicals are highly reactive and readily react with other substances. another difference is that ions are usually formed as a result of an atom or molecule gaining or losing.

Differentiate Between Ion And Free Radical Chy9chp1 Fundamentals Of Free radical: highly reactive due to the presence of an unpaired electron, seeking to pair it, which can lead to chain reactions. ion: its reactivity depends on the charge and the desire to return to a neutral state. ions can be relatively more stable than free radicals. One of the main differences between ions and free radicals is their stability. ions are generally stable and do not readily react with other substances, while free radicals are highly reactive and readily react with other substances. another difference is that ions are usually formed as a result of an atom or molecule gaining or losing. The main difference between a free radical and an ion lies in the number of unpaired electrons and their charge: free radicals: these are molecules or ions containing at least one unpaired electron. free radicals have one or more unpaired electrons, making them highly reactive. 15.1: free radical halogenation of alkanes. alkanes (the most basic of all organic compounds) undergo very few reactions. one of these reactions is halogenation, or the substitution of a single hydrogen on the alkane for a single halogen to form a haloalkane. this reaction is very important in organic chemistry because it opens a gateway to.

Difference Between Free Radical And Ionic Polymerization Compare The The main difference between a free radical and an ion lies in the number of unpaired electrons and their charge: free radicals: these are molecules or ions containing at least one unpaired electron. free radicals have one or more unpaired electrons, making them highly reactive. 15.1: free radical halogenation of alkanes. alkanes (the most basic of all organic compounds) undergo very few reactions. one of these reactions is halogenation, or the substitution of a single hydrogen on the alkane for a single halogen to form a haloalkane. this reaction is very important in organic chemistry because it opens a gateway to.

юааfreeюаб юааradicalюаб юааvsюаб юааionюаб таф Whatтащs юааthe Differenceюаб

Difference Between Ion And Free Radical Ion And Free Ra
