
Classification Of Alcohol Phenol And Ether Structure Naming Types Alcohols and phones are classified on the number of hydroxyl groups attached, monohydric alcohol contains one hydroxyl group, dihydric alcohol contains two hydroxyl groups and trihydric alcohol contains three hydroxyl groups. learn about the other classification of alcohol, phenol and ether primary, secondary and tertiary, symmetric and asymmetric with examples and structure. visit byju's for. Alcohol is the product we get when a saturated carbon atom bonds to a hydroxyl ( oh) group. phenol is what we get when the oh group replaces the hydrogen atom in benzene. ether is the product that we get when an oxygen atom bonds to two alkyl or aryl groups. browse more topics under alcohols phenols and ethers.

Alcohol Phenol And Ether Classification Types Videos 3,3 dibromo 2 methyl 2 butanol; tertiary. 6.1: alcohols nomenclature and classification is shared under a cc by nc sa 4.0 license and was authored, remixed, and or curated by libretexts. in the iupac system, alcohols are named by changing the ending of the parent alkane name to ol. alcohols are classified according to the number of carbon. Identify the following compounds as alcohol, phenol or ether. a. exercise \(\pageindex{2}\): the structure shown has a c h subscript 3 group bonded up and to the right to a c atom. the c atom is bonded down and to the right to a c h subscript 2 group. the c h subscript 2 group is bonded up and to the right to a c h subscript 2 group. For example, ch3oh is methyl alcohol. 7.1.2 phenols— mono, di and trihydric phenols ethers are classified as simple or symmetrical, if the alkyl or aryl groups attached to the oxygen atom are the same, and mixed or unsymmetrical, if the two groups are different. diethyl ether, c2h5oc2h5, is a symmetrical ether whereas c2h5och3 and c2h5oc6h5. Physical properties: (a) boiling point of ethers are much lower than corresponding alcohols because ethers do not form intermolecular h bonding. (b) slightly soluble in water. 10. chemical properties:(a) cleavage ofc o bond in ethers:(b) electrophilic substitution: in this, the alkoxy group activates the aromatic ring and directs the incoming.
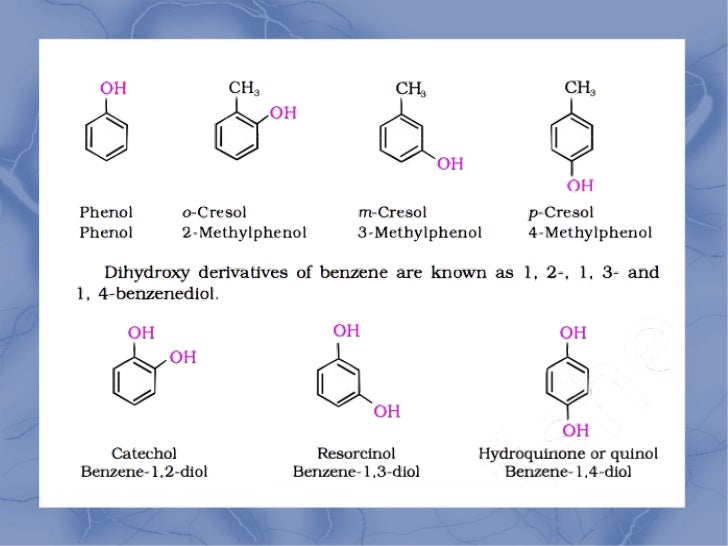
Classification Of Alcohol Phenol And Ether Structure Vrogue Co For example, ch3oh is methyl alcohol. 7.1.2 phenols— mono, di and trihydric phenols ethers are classified as simple or symmetrical, if the alkyl or aryl groups attached to the oxygen atom are the same, and mixed or unsymmetrical, if the two groups are different. diethyl ether, c2h5oc2h5, is a symmetrical ether whereas c2h5och3 and c2h5oc6h5. Physical properties: (a) boiling point of ethers are much lower than corresponding alcohols because ethers do not form intermolecular h bonding. (b) slightly soluble in water. 10. chemical properties:(a) cleavage ofc o bond in ethers:(b) electrophilic substitution: in this, the alkoxy group activates the aromatic ring and directs the incoming. Glycerol is the most important trihydroxy alcohol. 6.6: phenols phenols are compounds in which an oh group is attached directly to an aromatic ring. many phenols are used as antiseptics. 6.7: ethers to give ethers common names, simply name the groups attached to the oxygen atom, followed by the generic name ether. Check batch here: physicswallah.onelink.me zazb yt2june📲 pw app website: physicswallah.onelink.me zazb pwappweb📚 pw store: physicsw.

Alcohols Phenols And Ethers Classification Properties Nomenclature Glycerol is the most important trihydroxy alcohol. 6.6: phenols phenols are compounds in which an oh group is attached directly to an aromatic ring. many phenols are used as antiseptics. 6.7: ethers to give ethers common names, simply name the groups attached to the oxygen atom, followed by the generic name ether. Check batch here: physicswallah.onelink.me zazb yt2june📲 pw app website: physicswallah.onelink.me zazb pwappweb📚 pw store: physicsw.

Classification Of Alcohol Phenol And Ether

Solution Chm 232 Alcohols Phenols And Ethers Classification Of
