.jpg)
Ch4 Polar Or Nonpolar Covalent Bond Ppt The Chemistry Vrogue Co Ch4 is a nonpolar molecule as it has a symmetric tetrahedral geometrical shape with four identical c h bonds. the electronegativity of carbon and hydrogen is 2.55 and 2.2, respectively, which causes the partial charges to be almost zero. the difference in electrostatic potential is also minimal giving an overall nonpolar molecule. Methane (ch4) is a non polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms. methane is non polar as the difference in electronegativities between carbon and hydrogen is not great enough to form a polarized chemical bond. the Δen of carbon and hydrogen is ~0.35, too weak to be considered a true polar bond. as methane is non polar, it.

Ch4 Polar Or Nonpolar Covalent Bond Ppt The Chemistry Vrogue Co All the outer atoms are the same the same dipoles, and that the dipole moments are in the same direction towards the carbon atom, the overall molecule becomes non polar. therefore, methane has non polar bonds, and is non polar overall. keep in mind that molecules can have polar bonds, but be non polar overall. hope this helps :). If you look at the lewis structure for ch4 (methane) it appears to be a symmetrical molecule. however, to determine if ch4 is polar we consider the molecula. 5.10: electronegativity and bond polarity is shared under a not declared license and was authored, remixed, and or curated by libretexts. covalent bonds can be nonpolar or polar, depending on the electronegativities of the atoms involved. covalent bonds can be broken if energy is added to a molecule. the formation of covalent bonds is …. Let’s dive into it! ch4 (or methane) is a nonpolar molecule because all the four bonds (c h bonds) are identical and ch4 has symmetrical geometry. also the electronegativity difference of carbon atom (c = 2.55) and hydrogen atom (h = 2.2) is 0.35 (i.e 2.55 – 2.2 = 0.35), which is very less. hence the ch4 molecule is a nonpolar molecule.

Ch4 Polar Or Nonpolar Bonds Is Ch4 Polar Or Nonpolar Th 5.10: electronegativity and bond polarity is shared under a not declared license and was authored, remixed, and or curated by libretexts. covalent bonds can be nonpolar or polar, depending on the electronegativities of the atoms involved. covalent bonds can be broken if energy is added to a molecule. the formation of covalent bonds is …. Let’s dive into it! ch4 (or methane) is a nonpolar molecule because all the four bonds (c h bonds) are identical and ch4 has symmetrical geometry. also the electronegativity difference of carbon atom (c = 2.55) and hydrogen atom (h = 2.2) is 0.35 (i.e 2.55 – 2.2 = 0.35), which is very less. hence the ch4 molecule is a nonpolar molecule. 1) is called a nonpolar covalent bond. figure 4.4.1 4.4. 1 polar versus nonpolar covalent bonds. (a) the electrons in the covalent bond are equally shared by both hydrogen atoms. this is a nonpolar covalent bond. (b) the fluorine atom attracts the electrons in the bond more than the hydrogen atom does, leading to an imbalance in the electron. All the bonds in methane are the same (and are fairly non polar to start with). so, even if methane were flat with bonds forming a square with carbon at the centre, it would be non polar. it isn't flat: it is tetrahedral but, since all the bonds are the same, even a tiny dipole on each would still cancel out. carbon tetrachloride has 4 polar c.

Ch4 Polar Or Nonpolar Covalent Bond Name The Bond Pre Vrogue Co 1) is called a nonpolar covalent bond. figure 4.4.1 4.4. 1 polar versus nonpolar covalent bonds. (a) the electrons in the covalent bond are equally shared by both hydrogen atoms. this is a nonpolar covalent bond. (b) the fluorine atom attracts the electrons in the bond more than the hydrogen atom does, leading to an imbalance in the electron. All the bonds in methane are the same (and are fairly non polar to start with). so, even if methane were flat with bonds forming a square with carbon at the centre, it would be non polar. it isn't flat: it is tetrahedral but, since all the bonds are the same, even a tiny dipole on each would still cancel out. carbon tetrachloride has 4 polar c.

Ch4 Lewis Structure Molecular Geometry Polar Or Nonpolar Bond Angle
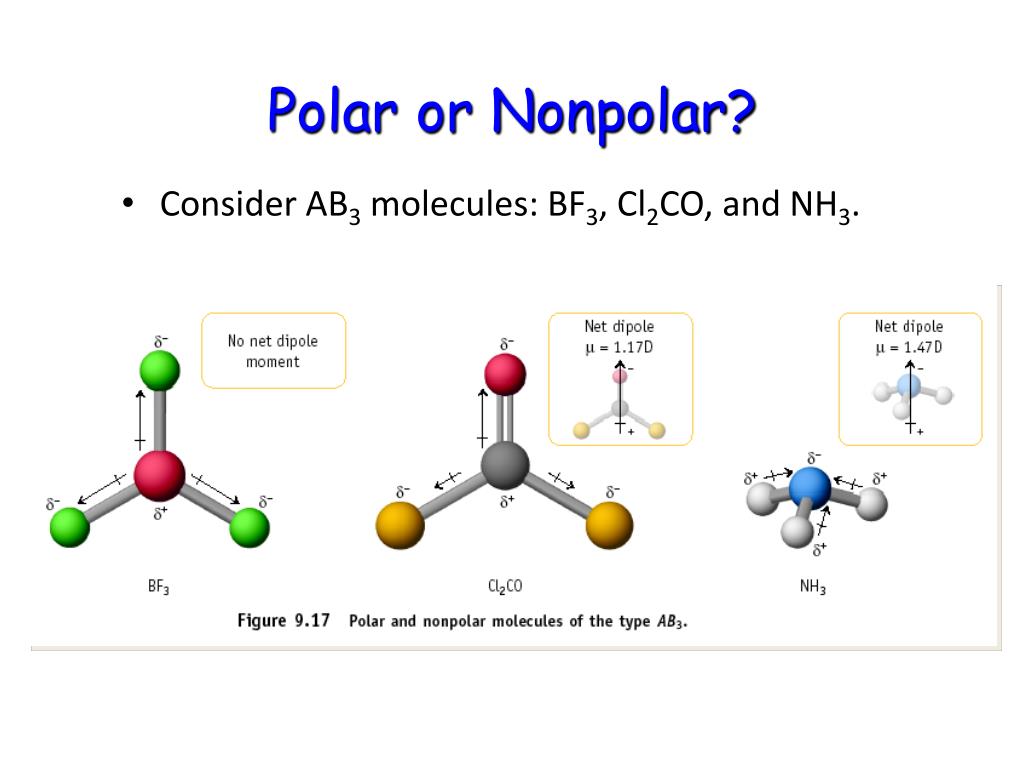
Ch4 Polar Or Nonpolar Covalent Bond Name The Bond Pre Vrogue Co
