
Ch4 Lewis Structure Molecular Geometry And Hybridization The lewis structure of the methane (ch4) molecule is drawn with four single shared covalent bonds between the carbon and hydrogen atoms each. moreover, as there exist sigma bonds only and one 2s and three 2p orbitals of the carbon produce four new hybrid orbitals, the hybridization of ch4 is sp3. it is interesting to realize that irrespective. Hence, ch 4 or methane has a tetrahedral molecular geometry. ch4 bond angles. one can use axn notation to find out the molecular geometry and the bond angles for any molecule. here ch4 follows the ax4 notation, and hence according to the table given below, the bond angles are 109.5° the ch 4 molecule will have 109.5° bond angles as there is.
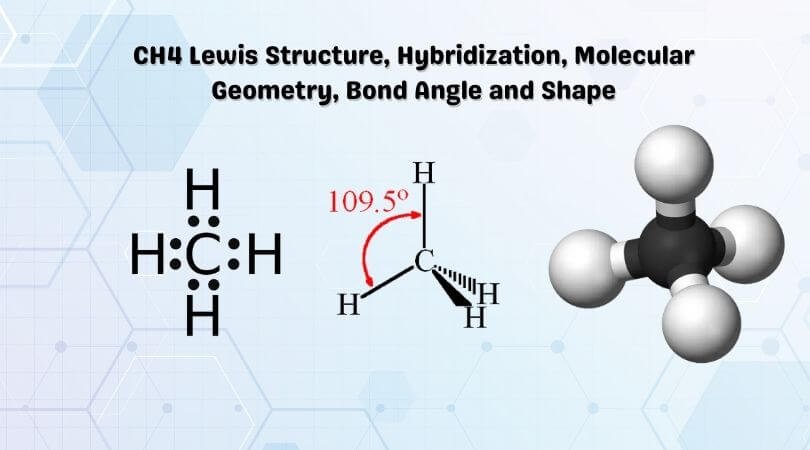
Ch4 Lewis Structure Hybridization Molecular Geometry Bond Angle A quick explanation of the molecular geometry for ch4 including bond angle, hybridization, and polarity of ch4.helpful resources:• how to draw lewis structur. The covalent bond in molecular fluorine, f 2, is a σ bond formed by the overlap of two half filled 2 p orbitals, one from each fluorine atom as shown here. figure 1.6f f2 σ bond. however, when the valence bond theory is applied to organic molecules, for instance ch 4, it does not work. the valence electron configuration of carbon atom is 2s 2. In order to understand the hybridization of ch 4 (methane), we have to take a look at the atomic orbitals which are of different shape and energy that take part in the process. the type of hybridization involved with ch4 is sp 3. we will discuss in detail how this hybridization occurs below. name of the molecule. methane. molecular formula. ch 4. Shapes of molecules and hybridization a. molecular geometry • lewis structures provide us with the number and types of bonds around a central atom, as well as any nb electron pairs. they do not tell us the 3 d structure of the molecule. h c h h h ch 4 as drawn conveys no 3 d information (bonds appear like they are 90° apart).

Ch4 Lewis Structure Molecular Geometry And Hybridization In order to understand the hybridization of ch 4 (methane), we have to take a look at the atomic orbitals which are of different shape and energy that take part in the process. the type of hybridization involved with ch4 is sp 3. we will discuss in detail how this hybridization occurs below. name of the molecule. methane. molecular formula. ch 4. Shapes of molecules and hybridization a. molecular geometry • lewis structures provide us with the number and types of bonds around a central atom, as well as any nb electron pairs. they do not tell us the 3 d structure of the molecule. h c h h h ch 4 as drawn conveys no 3 d information (bonds appear like they are 90° apart). A molecule of methane, ch 4, consists of a carbon atom surrounded by four hydrogen atoms at the corners of a tetrahedron. the carbon atom in methane exhibits sp 3 hybridization. we illustrate the orbitals and electron distribution in an isolated carbon atom and in the bonded atom in ch 4 in figure 8.16. the four valence electrons of the carbon. A good example is methane (ch 4). according to valence bond theory, the structure of a covalent species can be depicted using a lewis structure. experimentally, it has been shown that the four carbon hydrogen bonds in the methane molecule are identical, meaning they have the same bond energy and the same bond length.

Ch4 Lewis Structure Molecular Geometry And Hybridization A molecule of methane, ch 4, consists of a carbon atom surrounded by four hydrogen atoms at the corners of a tetrahedron. the carbon atom in methane exhibits sp 3 hybridization. we illustrate the orbitals and electron distribution in an isolated carbon atom and in the bonded atom in ch 4 in figure 8.16. the four valence electrons of the carbon. A good example is methane (ch 4). according to valence bond theory, the structure of a covalent species can be depicted using a lewis structure. experimentally, it has been shown that the four carbon hydrogen bonds in the methane molecule are identical, meaning they have the same bond energy and the same bond length.
