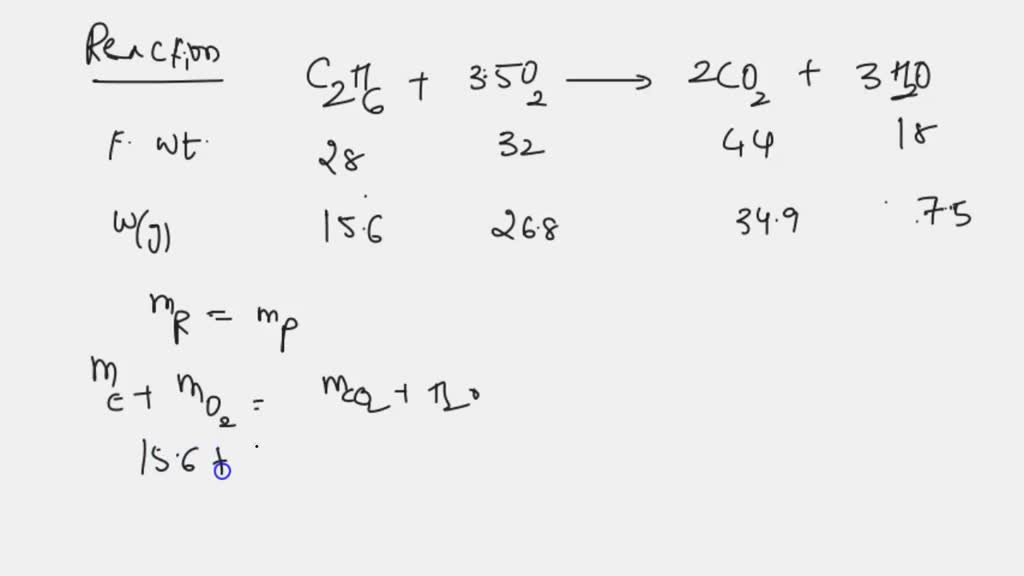
Solved Gaseous Ethane Ch3ch3 Reacts With Gaseous Oxygen Gas O2 To Step 4: substitute coefficients and verify result. count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges ions) are balanced. 2 ch3ch3 7 o2 = 4 co2 6 h2o. reactants. Word equation. propane dioxygen = carbon dioxide water. ch3(ch2)ch3 o2 = co2 h2o is a combustion reaction where one mole of propane [ch 3 (ch 2)ch 3] and five moles of dioxygen [o 2] react to form three moles of carbon dioxide [co 2] and four moles of water [h 2 o].

How To Balance Ch3ch3 O2 Co2 H2o Youtube Ch3 ch3 o2=co2 h2o balanced equation||ethane oxygen=carbon dioxide water balanced equationhow to balance ch3ch3 o2 = co2 h2o, how to balance ethane oxy. First, we set all coefficients to 1: 1 ch 3 (ch 2) 4 ch 3 1 o 2 = 1 co 2 1 h 2 o. for each element, we check if the number of atoms is balanced on both sides of the equation. c is not balanced: 6 atoms in reagents and 1 atom in products. in order to balance c on both sides we: multiply coefficient for co 2 by 6. Stoichiometric balanced chemical reaction of ethane and oxygen gases. two moles of ethane reacts with seven moles of oxygen gas and produce four moles of carbon dioxide and six moles of water. 2ch 3 ch 3(g) 7o 2(g) → 4co 2 6h 2 o. heat generation. as methane, ethane can be used for heat generation from the combustion with oxygen gas. The combustion reaction calculator will give you the balanced reaction for the combustion of hydrocarbons or c, h, o substances. to use the calculator, enter the molecular formula of your substance: on the first row, total atoms of carbon c (α), enter the number of carbon atoms of your substance. then, on the total atoms of hydrogen h (β.
CH3+%2B+O2+%3D+CO2+%2B+H2O)
Ch3 Ch2 Ch3 O2 Co2 H2o Chemical Equation Balancer Stoichiometric balanced chemical reaction of ethane and oxygen gases. two moles of ethane reacts with seven moles of oxygen gas and produce four moles of carbon dioxide and six moles of water. 2ch 3 ch 3(g) 7o 2(g) → 4co 2 6h 2 o. heat generation. as methane, ethane can be used for heat generation from the combustion with oxygen gas. The combustion reaction calculator will give you the balanced reaction for the combustion of hydrocarbons or c, h, o substances. to use the calculator, enter the molecular formula of your substance: on the first row, total atoms of carbon c (α), enter the number of carbon atoms of your substance. then, on the total atoms of hydrogen h (β. Gaseous ethane (ch3ch3) will react with gaseous oxygen (o2) to produce gaseous carbon dioxide (co2) and gaseous water (h2o) suppose 27.4 g of ethane is mixed with 42. g of oxygen. calculate the maximum mass of water that could be produced by the chemical reaction. Balanced equation. find limiting reactant. an easy way is to simply divide moles of each reactant by the corresponding coefficient in the balanced equation. whichever value comes out less is the limiting reactant. for c 4 h 10: 4.65 g c 4 h 10 x 1 mol 58.1 g = 0.0800 moles c 4 h 10 (÷2 >0.04) for o 2: 26.5 g o 2 x 1 mol o 2 32 g = 0.828.
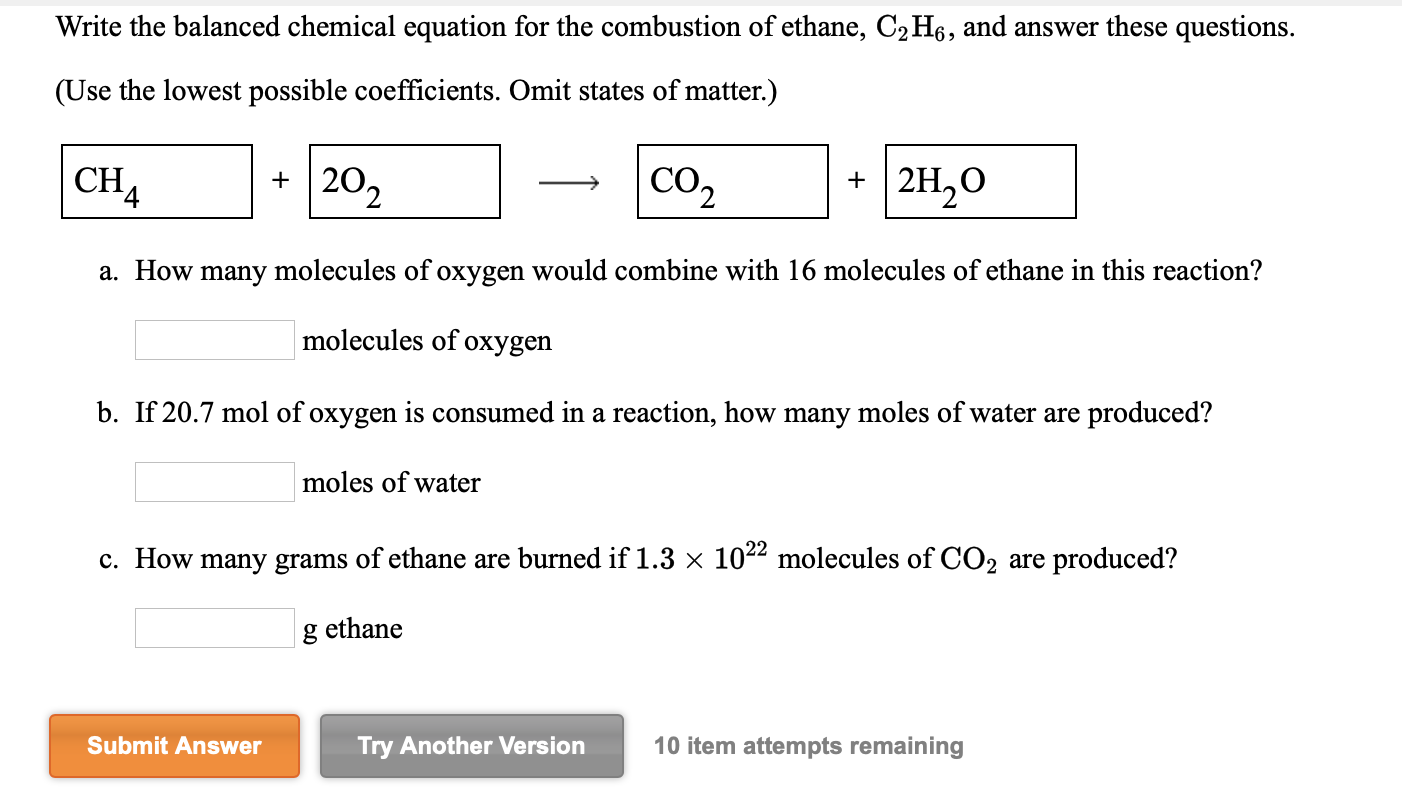
Solved Write The Balanced Chemical Equation For The Chegg Gaseous ethane (ch3ch3) will react with gaseous oxygen (o2) to produce gaseous carbon dioxide (co2) and gaseous water (h2o) suppose 27.4 g of ethane is mixed with 42. g of oxygen. calculate the maximum mass of water that could be produced by the chemical reaction. Balanced equation. find limiting reactant. an easy way is to simply divide moles of each reactant by the corresponding coefficient in the balanced equation. whichever value comes out less is the limiting reactant. for c 4 h 10: 4.65 g c 4 h 10 x 1 mol 58.1 g = 0.0800 moles c 4 h 10 (÷2 >0.04) for o 2: 26.5 g o 2 x 1 mol o 2 32 g = 0.828.
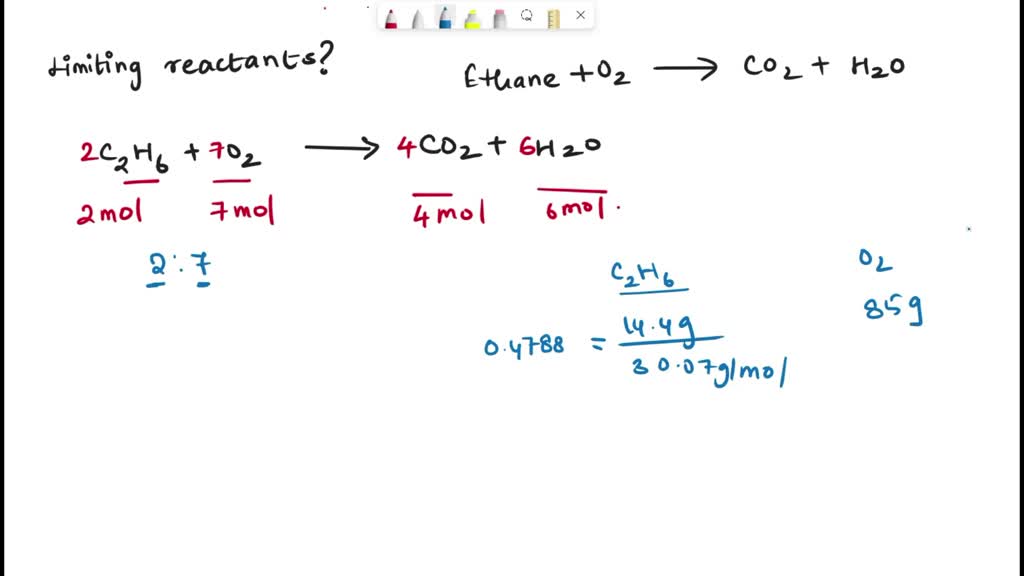
Solved Limiting Reactant Gaseous Ethane Ch3ch3 Will React With Gaseous

Balanced Equation Ethane And Oxygen Reaction To Form Co And H2o
