
Ash3 Lewis Structure Arsenic Trihydride Youtube A step by step explanation of how to draw the ash3 lewis dot structure (arsenic trihydride).for the ash3 structure use the periodic table to find the total n. Hello everyone! welcome back to our channel, and for today’s video, we will help you determine the lewis structure of the ash3 molecule. to find out this str.

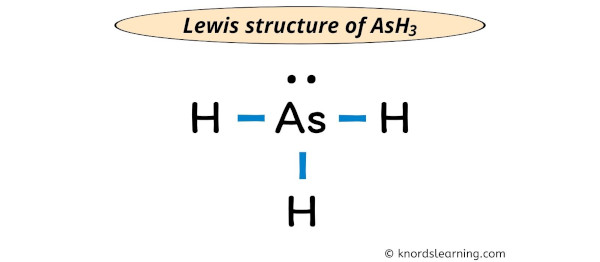
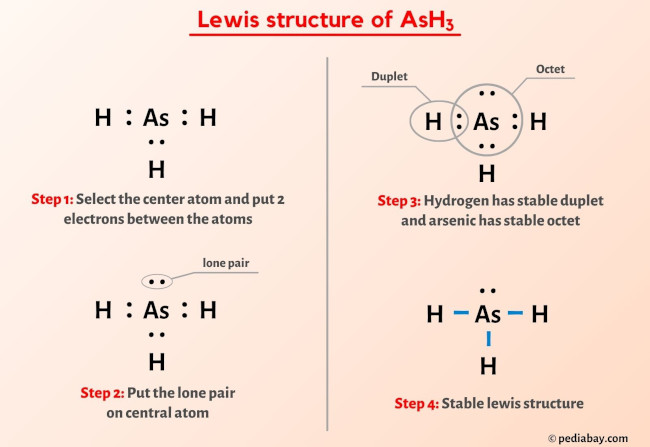
Ash3 Lewis Structure How To Draw The Lewis Dot Structure о A step by step explanation of how to draw the arsenic (as) lewis dot structure.for the arseniclewis structure use the periodic table to find the total number. The arsenic atom (as) is at the center and it is surrounded by 3 hydrogen atoms (h). the arsenic atom has one lone pair. let’s draw and understand this lewis dot structure step by step. (note: take a pen and paper with you and try to draw this lewis structure along with me. i am sure you will definitely learn how to draw lewis structure of ash3). Transcript: this is the ash3 lewis structure: arsenic trihydride. as goes at the center, hydrogens always on the outside. we have three of them. for the ash3 lewis structure, we have 5 valence electrons for as, plus 1 for hydrogen, but we have 3 hydrogens, for a total of 5 3 8 valence electrons. we'll put 2 valence electrons between the atoms. Steps to draw the lewis structure of ash3. step 1. count the total number of valence shell electrons on the compound. this is done by adding the valence shell electrons of all the constituent atoms. atom. atomic number. group number. valence electrons according to group number. electronic configuration (e.c.).

Lewis Structure Of Ash3 With 6 Simple Steps To Draw Vrogue Co Transcript: this is the ash3 lewis structure: arsenic trihydride. as goes at the center, hydrogens always on the outside. we have three of them. for the ash3 lewis structure, we have 5 valence electrons for as, plus 1 for hydrogen, but we have 3 hydrogens, for a total of 5 3 8 valence electrons. we'll put 2 valence electrons between the atoms. Steps to draw the lewis structure of ash3. step 1. count the total number of valence shell electrons on the compound. this is done by adding the valence shell electrons of all the constituent atoms. atom. atomic number. group number. valence electrons according to group number. electronic configuration (e.c.). Step 3: connect each atoms by putting an electron pair between them. now in the ash3 molecule, you have to put the electron pairs between the arsenic atom (as) and hydrogen atoms (h). this indicates that the arsenic (as) and hydrogen (h) are chemically bonded with each other in a ash3 molecule. step 4: make the outer atoms stable. Molecular geometry of arsenic . the lewis structure implies that arsenic trihydride (ash3) adopts a trigonal pyramidal geometry. in this arrangement, the three hydrogen atoms are positioned asymmetrically around the central arsenic atom, forming three bond pairs and one lone pair.

Ash3 Lewis Structure Geometry Hybridization And Polarity Step 3: connect each atoms by putting an electron pair between them. now in the ash3 molecule, you have to put the electron pairs between the arsenic atom (as) and hydrogen atoms (h). this indicates that the arsenic (as) and hydrogen (h) are chemically bonded with each other in a ash3 molecule. step 4: make the outer atoms stable. Molecular geometry of arsenic . the lewis structure implies that arsenic trihydride (ash3) adopts a trigonal pyramidal geometry. in this arrangement, the three hydrogen atoms are positioned asymmetrically around the central arsenic atom, forming three bond pairs and one lone pair.
Lewis Structure Of Ash3 With 6 Simple Steps To Draw
Ash3 Lewis Structure In 6 Steps With Images
