
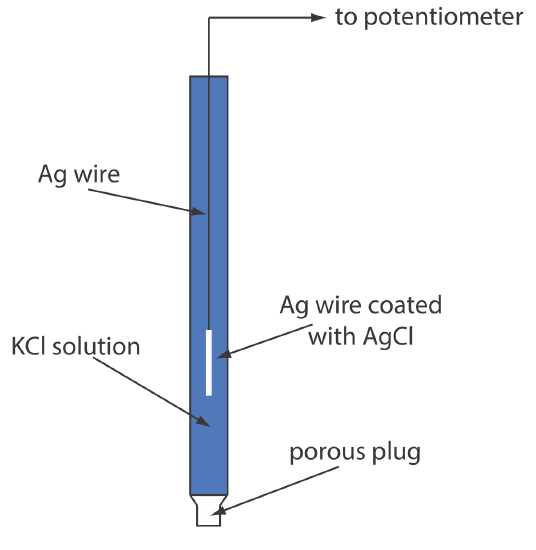
Schematic Cross Section Of A Simple Ag Agcl Reference Electrode Another common ag agcl electrode uses a solution of 3.5 m kcl and has a potential of 0.205 v at 25 o c. as you might expect, the potential of a ag agcl electrode using a saturated solution of kcl is more sensitive to a change in temperature than an electrode that uses an unsaturated solution of kcl. a typical ag agcl electrode is shown in. 2. silver silver chloride (ag agcl) the silver silver chloride reference electrode is composed of a silver wire, sometimes coated with a layer of solid silver chloride, immersed in a solution that is saturated with potassium chloride and silver chloride. the pertinent half reaction is. agcl(s) e − ⇔ ag(s) cl − (sat’d) with a value.

A Structure Of Ag Agcl Reference Electrode Download Scientific Diagram Section 4. reference electrodes. 26. if the two electrodes are of the same type (e.g., ag agcl vs. ag agcl, or calomel vs. calomel) the meter should read 0 ±20 mv. if your reading for any pair of electrodes is significantly different, you should have another electrode of the same type handy to help distinguish which of the two is bad. The ag agcl reference electrode is a popular and widely used electrode in electrochemical measurements. its structure is simple, yet effective, consisting of a silver wire coated with a layer of silver chloride. the electrode works based on the redox reaction between ag and agcl, which acts as a reversible redox couple. Commercial reference electrodes consist of a glass or plastic tube electrode body. the electrode consists of a metallic silver wire (ag (s)) coated with a thin layer of silver chloride (agcl), either physically by dipping the wire in molten silver chloride, chemically by electroplating the wire in concentrated hydrochloric acid (hcl) [3] or electrochemically by oxidising the silver at an anode. \[ag(s) cl^–(aq) →agcl(s) e^–\] this electrode usually takes the form of a piece of silver wire coated with agcl. the coating is done by making the silver the anode in an electrolytic cell containing hcl; the ag ions combine with cl – ions as fast as they are formed at the silver surface. the other common reference electrode is.
23 1 Reference Electrodes Chemistry Libretexts Commercial reference electrodes consist of a glass or plastic tube electrode body. the electrode consists of a metallic silver wire (ag (s)) coated with a thin layer of silver chloride (agcl), either physically by dipping the wire in molten silver chloride, chemically by electroplating the wire in concentrated hydrochloric acid (hcl) [3] or electrochemically by oxidising the silver at an anode. \[ag(s) cl^–(aq) →agcl(s) e^–\] this electrode usually takes the form of a piece of silver wire coated with agcl. the coating is done by making the silver the anode in an electrolytic cell containing hcl; the ag ions combine with cl – ions as fast as they are formed at the silver surface. the other common reference electrode is. A reference electrode is an electrode that has a stable and well known electrode potential. the overall chemical reaction taking place in a cell is made up of two independent half reactions, which describe chemical changes at the two electrodes. to focus on the reaction at the working electrode, the reference electrode is standardized with. The ag agcl reference electrode is a commonly used reference electrode in electrochemistry; however, because of the size mismatch problem, pseudo reference electrodes are typically used in microfluidic systems to provide liquid gating and stable solution potential (chen et al., 2009; 2011).

The Potential Of The Ag Agcl Electrode With Various Concentration A reference electrode is an electrode that has a stable and well known electrode potential. the overall chemical reaction taking place in a cell is made up of two independent half reactions, which describe chemical changes at the two electrodes. to focus on the reaction at the working electrode, the reference electrode is standardized with. The ag agcl reference electrode is a commonly used reference electrode in electrochemistry; however, because of the size mismatch problem, pseudo reference electrodes are typically used in microfluidic systems to provide liquid gating and stable solution potential (chen et al., 2009; 2011).

Construction Of A Typical Screen Printed Ag Agcl Reference Electrode

Figure 1 From Stability And Placement Of Ag Agcl Quasi Reference
